In the presence of a special type of catalyst, hydrogen gas will add across a triple bond
Question:
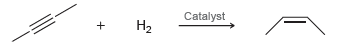
The process is exothermic. Do you expect a high temperature to favor products or reactants?
Transcribed Image Text:
H2 Catalyst
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (13 reviews)
In order to determine if reactants or products are favored at high temperature we must cons...View the full answer

Answered By

Shubhradeep Maity
I am an experienced and talented freelance writer passionate about creating high-quality content. I have over five years of experience working in the field and have collaborated with several renowned companies and clients in the SaaS industry.
At Herman LLC, an online collective of writers, I generated 1,000+ views on my content and created journal content for 100+ clients on finance topics. My efforts led to a 60% increase in customer engagement for finance clients through revamping website pages and email interaction.
Previously, at Gerhold, a data management platform using blockchain, I wrote and published over 50 articles on topics such as Business Finance, Scalability, and Financial Security. I managed four writing projects concurrently and increased the average salary per page from $4 to $7 in three months.
In my previous role at Bernier, I created content for 40+ clients within the finance industry, increasing sales by up to 40%.
I am an accomplished writer with a track record of delivering high-quality content on time and within budget. I am dedicated to helping my clients achieve their goals and providing exceptional results.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The synthesis of ethyl chloride is accomplished by reacting ethylene with hydrogen chloride in the presence of an aluminum chloride catalyst: C 2 H 4 (g) + HC1 (g) catalyst ? C 2 H 5 C1 (g): ?H ?...
-
Caryophyllene, a compound found in oil of cloves, has the molecular formula C15H24 and has no triple bonds. Reaction of caryophyllene with an excess of hydrogen in the presence of a platinum catalyst...
-
When ethene is mixed with hydrogen in the presence of a platinum catalyst, hydrogen adds across the double bond to form ethane. At room temperature, the reaction goes to completion. Predict the signs...
-
Due to acid rain, the percentage of lakes in Scandinavia that lost their population of brown trout increased dramatically between 1940 and 1975. Based on a sample of 2850 lakes, this percentage can...
-
In terms of ICS/SCADA risk management and auditing, demonstrate how to use your knowledge of the risk as function of M, AV, T, and V. That is R = f (M, AV, T, V). Where: R - Risk, M - Mission...
-
Derive an equation for the ratio of tension in the belt drive.
-
The fiscal year of the Eddy Company ends on December 31. Your audit report, dated February 26, is to be delivered to the client on March 9. Listed below are events that occur or are discovered from...
-
Mandy Knox is the new manager of the materials storeroom for Timken Manufacturing. Mandy has been asked to estimate future monthly purchase costs for part # 696, used in two of Timkens products....
-
A particle of dust lands 46.4 mm from the center of a compact disc (CD) that is 120 mm in diameter. The CD speeds up from rest, and the dust particle is ejected when the CD is rotating at 96.0...
-
Book Nook Warehouse distributes hardcover books to retail stores and extends credit terms of 2/10, n/30 to all of its customers. At the end of May, Book Nook's inventory consisted of books purchased...
-
Identify whether each of the following factors will affect the rate of a reaction: (a) K eq (b) G (c) Temperature (d) H (e) E a (f ) S
-
Consider the following reaction. Predict whether an increase in temperature will favor reactants or products. Justify your prediction. +
-
In Exercises 2132, find the domain and range of the function. Z = 2 AX x + y
-
write a perfect body paragraph on topic immigration experience to Canada in the following format Main idea Supporting detail 1 Evidence Supporting detail 2 Evidence Conclusion sentence.
-
Using the selected emotionally incompetent behavior: Displaying a bad attitude. Please address the following. 1. Develop a fictitious example that demonstrates the selected behavior. 2. Using the...
-
Royal Oaks Realty's net revenue and net income for the following five-year period, using 2021 as the base year, follow: (Click the icon to view net revenue and net income for the five years.)...
-
How can one describe the relationship between Cassatt and Degas, given the ways they interacted with each other ?
-
Assume that Schmidt Machinery Company had the standard costs reflected in Exhibit 14.5. In a given month, the company used 3,475 pounds of aluminum to manufacture 925 units. The company paid $29.00...
-
Find (a) the curl and (b) the divergence of the vector field. F(x, y, z) = xye z i + yze x k
-
Read Case Study Google: Dont Be Evil Unless and answer the following: Given its mission of providing information to the world, should Google censor searches in China?
-
Using a different method for each part, but taking care in each case to select a good method, show how each of the following transformations might be accomplished: (a) (b) (c) (d) (e) NH2 NH2 CH3O...
-
Review the chemistry of amines given in earlier sections and provide a specific example for each of the previously illustrated reactions.
-
Para-nitrosation of N,N-dimethylaniline (C-nitrosation) is believed to take place through an electrophilic attack by N+O ions. (a) Show how N+O ions might be formed in an aqueous solution of NaNO 2...
-
Find 7e dx + C
-
Explain the differences between containerization and virtualization. What are the benefits and limitations of each approach in terms of resource isolation and scalability ?
-
Discuss common techniques used to optimize operating system performance. How do profiling, caching, and load balancing play a role in improving system efficiency ?

Study smarter with the SolutionInn App


