Consider the following reaction. Predict whether an increase in temperature will favor reactants or products. Justify your
Question:
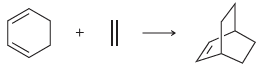
Transcribed Image Text:
+
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 87% (16 reviews)
Recall that G has two components H and TS We must analyze each term separately The first term is exp...View the full answer

Answered By

Cyrus Sandoval
I a web and systems developer with a vast array of knowledge in many different front end and back end languages, responsive frameworks, databases, and best code practices. My objective is simply to be the best web developer that i can be and to contribute to the technology industry all that i know and i can do. My skills include:
- Front end languages: css, HTML, Javascript, XML
- Frameworks: Angular, Jquery, Bootstrap, Jasmine, Mocha
- Back End Languages: Java, Javascript, PHP,kotlin
- Databases: MySQL, PostegreSQL, Mongo, Cassandra
- Tools: Atom, Aptana, Eclipse, Android Studio, Notepad++, Netbeans.
Having a degree in Computer Science enabled me to deeply learn most of the things regarding programming, and i believe that my understanding of problem solving and complex algorithms are also skills that have and will continue to contribute to my overall success as a developer.
I’ve worked on countless freelance projects and have been involved with a handful of notable startups. Also while freelancing I was involved in doing other IT tasks requiring the use of computers from working with data, content creation and transcription.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the following reaction to produce methyl acetate: When this reaction is carried out with CH3OH containing radioactive oxygen-18, the water produced is not radioactive. Explain. CH,OH CH...
-
Consider the following reaction at some temperature: H2O(g) + CO(g) H2(g) + CO2(g) K = 2.0 Some molecules of H2O and CO are placed in a 1.0- L container as shown below. When equilibrium is reached,...
-
Consider the following reaction at 248oC and 1.00 atm: CH3Cl(g) + H2(g) CH4(g) + HCl(g) For this reaction, the enthalpy change at 248oC is 283.3 kJ/ mol. At constant pressure the molar heat...
-
Suppose that one worker can shovel snow from a storefront sidewalk in 50 minutes and another worker can shovel it in 30 minutes. How long will it take if they work together?
-
1. Develop and submit the Mini-Case Just Right Globalization 2. Is there more to the "not-invented-here" syndrome than simply hurt feelings on the part of those who believe they are being dictated to...
-
Explain chain drives and rope drives, and their applications?
-
In connection with your examination of the financial statements of Olars Manufacturing Corporation for the year ended December 31, 19X0, your post-balance-sheet-date review disclosed the following...
-
Troy Industries purchased a new machine 3 years ago for $80,000. It is being depreciated under MACRS with a 5-year recovery period using the percentages given in Table 4.2. Assume a 40% tax rate. a....
-
You are a candidate attorney at a law firm specialising in child justice. You sit in in a consultation with your principal, where the client is the grandparent of a 14-year-old girl. The grandmother...
-
Prepare a balance sheet as of June 30, for the J. L. Gregory Company, using the following data: Accounts payable Accounts receivable Accrued expenses Accumulated depreciation 241,000 505,000 107,000...
-
In the presence of a special type of catalyst, hydrogen gas will add across a triple bond to produce a double bond: The process is exothermic. Do you expect a high temperature to favor products or...
-
When an amine is protonated, the resulting ammonium ion is not electrophilic: However, when an imine is protonated, the resulting iminium ion is highly electrophilic: Explain this difference in...
-
Canadian Tire has one of the oldest, most well-known loyalty programs in Canada, Canadian Tire money. Examine Notes 3 and 22 regarding customer loyalty and answer the following questions: a. Why do...
-
Comparative financial statements for Weller Corporation, a merchandising company, for the year ending December 31 appear below. The company did not issue any common stock during the year. A total of...
-
What interventions are the most beneficial to the clients your agency serves? Explain
-
Train-the-trainer manual for the training and development department within your organization or for one with which you are familiar. Your trainers will soon be responsible to train the staff about...
-
Find the limit 4 lim (x -2x32x + x 1) =? 2+1 -
-
According to the Hawail Wildlife Fund, North Pacific Humpback Whales migrate from the icy waters around Alaska during the fall to spend the winter in Hawaii where they mate, give birth, and nurture...
-
Find (a) the curl and (b) the divergence of the vector field. F(x, y, z) = x 3 yz 2 j + y 4 z 3 k
-
A certain Christmas tree ornament is a silver sphere having a diameter of 8.50 cm. Determine an object location for which the size of the reflected image is three-fourths the size of the object. Use...
-
If we examine Table 21.1, we find that the methylphenols (cresols) are less acidic than phenol itself. For example, This behavior is characteristic of phenols bearing electron-releasing groups....
-
When o-chlorotoluene is subjected to the conditions used in the Dow process (i.e., aqueous NaOH at 350oC at high pressure), the products of the reaction are o-cresol and m-cresol. What does this...
-
When 2-bromo-1, 3-dimethylbenzene is treated with sodium amide in liquid ammonia, no substitution takes place. This result can be interpreted as providing evidence for the elimination-addition...
-
(a) Argue that sin(x)dx = f sin(x)dx + fo sin(x)dx, and use this to compute a (b) Explain how to calculate where u, v, f are functions. (c) Let a >0. Calculate d da L sin(x)dx -a d v(a) f(x)dx da d...
-
= [(-1)* (2m)!" 12. Using Maclaurin series of cosx=(-1)*- A. Find Maclaurin series of cosx. B. Using the 1st four terms of Maclaurin series of cosx to evaluate focos x dx (leave answer to four...
-
Confirm that the below limit meets the conditions to apply l'Hpital's Rule and then solve the limit. Be sure to address these conditions in your explanation. Enter an exact numeric answer. lim In(x)...

Study smarter with the SolutionInn App


