Ixabepilone is a cytotoxic compound approved by the FDA in 2007 for the treatment of advanced breast
Question:
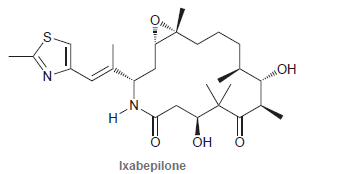
Transcribed Image Text:
он N. Он Ixabepilone
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
N ...View the full answer

Answered By

Allan Olal
I have vast tutoring experience of more than 8 years and my primary objective as a tutor is to ensure that a student achieves their academic goals.
4.70+
78+ Reviews
411+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
p53 gene therapy a. Triggers cytotoxic T cells to destroy tumor cells. b. Triggers apoptosis in cancer cells. c. Produces monoclonal antibodies against the tumor cells. d. Reduces tumors by breaking...
-
Describe the specific roles of helper, regulatory, and cytotoxic T cells in normal cellular immunity.
-
Compound A undergoes catalytic hydrogenation much faster than does compound B. Why? Making molecular models will help.
-
Graph the sets of points whose polar coordinates satisfy the equations and inequalitie. = 2/3, r -2
-
Here is the case "Marketing research centers: community, productivity, and relevance" Read that case carefully and write a summary about it.
-
The ten top-grossing Pixar animated movies for the U.S. box office up to June 2013 are shown in the table on the next page, in millions of dollars. a. Sort the gross income from smallest (on the...
-
A thin-walled filament-wound composite pressure vessel has fibers wound at a helical angle , as shown in Figure 1.45. Ignore the resin matrix material and assume that the fibers carry the entire...
-
On September 30, 2017, the Parker Corporation had a balance of $50,500 in its cash account. Additional information: Actual sales: August........................$180,000...
-
Determine the magnitude, direction and position of the force which will keep the body in equilibrium? 40 N 80 N 45% 1.5 m 30 1.5 m 1.5 m 30 1.5 m 45 80 N 60 N
-
The following are data regarding last years production of Dicer Ricer, one of the major products of Kitchen Gadget Company. During the year, 61,000 units of this product were manufactured and 62,100...
-
Draw the enantiomer of each of the following compounds: a. b. c. d. e. f. g. Albuterol (sold under the trade name VentolinTM) A bronchodilator used in the treatment of asthma N. Propranolol A...
-
Each of the following compounds possesses carbon atoms that are chirality centers. Locate each of these chirality centers, and identify the configuration of each one: a. b. c. d. e. f. Ephedrine A...
-
Youre driving along at 25 m/s with your aunts valuable antiques in the back of your pickup truck when suddenly you see a giant hole in the road 55 m ahead of you. Fortunately, your foot is right...
-
Burnout Oil Corporation uses the full cost method. Recently, the company acquired a truck costing $60,000 with an estimated life of five years (ignore salvage value). The foreman drives the truck to...
-
Using the information in this chapter, label each of the following statements true, false, or uncertain. Explain briefly. a. The saving rate is always equal to the investment rate. b. A higher...
-
Convergence between Japan and the United States since 1960 The Bureau of Labor Statistics has a user-friendly Web site of GDP per capita at...
-
RED and growth a. Why is the amount of R\&D spending important for growth? How do the appropriability and fertility of research affect the amount of R\&D spending? How do each of the policy proposals...
-
The following table shows data on asthma-related visits. Is there evidence that these visits vary by quarter? Can you detect a trend? A powerful test would be to run a multiple regression in Excel....
-
What is the effect of a subsidy in a market with a horizontal supply curve? With a vertical supply curve?
-
Use translations to graph f. f(x) = x-/2 +1
-
Outlined here is a synthesis of glyceraldehyde (Section 5.15A). What are the intermediates A-C and what stereoisomeric form of glyceraldehyde would you expect to obtain? PCC CH3OH, HA KMnO4, HO H2o ...
-
Consider the reduction of (R)-3-phenyl-2-pentanone by sodium borohydride. After the reduction is complete, the mixture is separated by chromatography into two fractions. These fractions contain...
-
The structure of the sex pheromone (attractant) of the female tsetse fly has been confirmed by the following synthesis. Compound C appears to be identical to the natural pheromone in all respects...
-
Vanessa has an outstanding bond with a 7.10 % semi-annual coupon and has 16 years left to mature. The current market rate is 10.50% per annum. What is the current yield of Vanessa's Bond.
-
Estimate the hydrogen consumption required to completely remove the sulfur from a hydrotreater feedstock and to reduce the nitrogen content of the product to 15 ppm by weight. The 48.5 API naphtha...
-
A firm has 36.30 million shares of common stock outstanding at a price of $32.81 per share. The firm also has 492000.00 bonds outstanding with a current price of $1,089.00. The outstanding bonds have...

Study smarter with the SolutionInn App


