Nicotine is a toxic substance present in tobacco leaves. There are two lone pairs in the structure
Question:
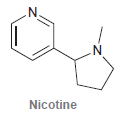
There are two lone pairs in the structure of nicotine. In general, localized lone pairs are much more reactive than delocalized lone pairs. With this information in mind, do you expect both lone pairs in nicotine to be reactive? Justify your answer.
Transcribed Image Text:
Nicotine
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (16 reviews)
Both lone pairs ...View the full answer

Answered By

Gilbert Chesire
I am a diligent writer who understands the writing conventions used in the industry and with the expertise to produce high quality papers at all times. I love to write plagiarism free work with which the grammar flows perfectly. I write both academics and articles with a lot of enthusiasm. I am always determined to put the interests of my customers before mine so as to build a cohesive environment where we can benefit from each other. I value all my clients and I pay them back by delivering the quality of work they yearn to get.
4.80+
14+ Reviews
49+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
White phosphorus is much more reactive than black or red phosphorus. Explain.
-
Why is trityl chloride much more reactive than the other alkyl halides in Table 17.2? TABLE 17 2 comparison of S,1 Solvolysis Rates of Benzylic and Nonbenzylic Alkyl Halides 25C R-CIHo l + H2O -OH +...
-
Ethyl triflate is much more reactive than ethyl mesylate toward nucleophiles in SN2 reactions. (a) Give the structures of all of the products formed when each compound reacts with potassium iodide in...
-
You bought a share of 3.4 percent preferred stock for $96.82 last year. The market price for your stock is now $98.34. What is your total return for last year?
-
This cases focuses on the issue of performance. As discussed in the text, clear and effective goals help improve employee performance, reduce role stress conflict and ambiguity, and improve the...
-
Sketch some equipotential lines for the electric field line pattern of an electric dipole.
-
Critically analyse how the external audit in the public sector differs from that in the private sector.
-
Lovvern Trophies makes and sells trophies it distributes to little league ballplayers. The company normally produces and sells between 8,000 and 14,000 trophies per year. The following cost data...
-
What is the difference between "equal pay for equal work" and "equal pay for work of equal value" and the implications and differences for the HR Manager?
-
On February 11, 20Y9, Quick Fix Company purchased $2,250 of supplies on account. In Quick Fixs chart of accounts, the supplies account is No. 15, and the accounts payable account is No. 21. a....
-
For each compound below, identify all lone pairs and indicate whether each lone pair is localized or delocalized. Then, use that information to determine the hybridization state and geometry for each...
-
A 455 cm 3 vessel contains a mixture of Ar and Xe. If the mass of the gas mixture is 2.245 g at 25.0C and the pressure is 760. Torr, calculate the mole fraction of Xe in the mixture.
-
In Problems 4576, solve each exponential equation. Express irrational solutions in exact form. ex - e-x 2 || 2
-
What performance metric of those listed below is the safest to use in order to determine performance of an organization and why? Explain. Net Income, Cash Flow From Operations, EBITDA
-
The GAAP regulations allow for variations in reporting financial data. What is the purpose of these options? There has also are been discussion to replace the GAAP with IFRSstandards.Discuss.
-
All financial institutions, to varying degrees, are exposed to credit, interest rate, liquidity, foreign exchange, and operating risks. Briefly and clearly define each.
-
Why are the liabilities and cash outlays of a property and casualty insurance company more difficult to predict than for a life insurance company?What are the effects of these differences on the P/C...
-
Hartley, Inc. needs to purchase equipment for its 2,000 drive-ins nationwide. The total cost of the equipment is $2 million. It is estimated that the aftertax cash infows from the project will be...
-
Let and let O be the 2 2 zero matrix. Show that each statement is true. For square matrices A and B of the same dimension, if AB = O and if A -1 exists, then B = O. d
-
Suppose that a flow network G = (V, E) violates the assumption that the network contains a path s t for all vertices V. Let u be a vertex for which there is no path s u t. Show that there must...
-
Make a reaction flowchart (roadmap diagram), as in previous problems, to organize the information provided to solve this problem. An optically active compound A (assume that it is dextrorotatory) has...
-
A naturally occurring antibiotic called mycomycin has the structure shown here. Mycomycin is optically active. Explain this by writing structures for the enantiomeric forms of mycomycin....
-
An optically active compound D has the molecular formula C6H10 and shows a peak at about 3300 cm-1 in its IR spectrum. On catalytic hydrogenation D yields E (C6H14). Compound E is optically inactive...
-
According to the video "The Importance of Budgeting," budgeting is a crucial aspect of running a successful business as it helps administrators allocate resources effectively to meet their goals. It...
-
Managing finances is essential for any business, big or small. It involves careful planning and allocation of resources to achieve goals. Budgeting is crucial as it helps firms manage their cash...
-
Your boss (for 1 of the six organizations you have to choose from-Google, Walt Disney Company, Amazon, P&G, 3M, or Ford Motor Company) has asked you to represent the organization in an industry panel...

Study smarter with the SolutionInn App


