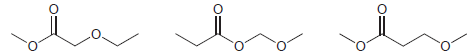
A 1 H NMR spectrum was acquired for each of the following constitutional isomers. Comparison of the
Question:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (7 reviews)
HH methylene proto...View the full answer

Answered By

Stephanie Olivero
No, Education & Tutoring Experience
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Identify one possible research problem for each of the following decision problems. Tell whether the decision problems are discovery-oriented or strategy-oriented. a. Why have sales of my brand...
-
Identify the best answer for each of the following 1. Which of the following statements concerning the accounting and financial reporting for capital assets is false? a. Capitalization thresholds...
-
Identify the location strategies for each of the following types of retailers. Department sorest Specialty apparel stores Category killers/specialists Grocery stores
-
Mario and Kaitlin are married and file a joint tax return. They have adjusted gross income of $385,000 that includes $4,700 of investment income ($3,000 short-term capital gains and $1,700 of...
-
Using the basic expectations theory, describe how the shape of the yield curve is determined.
-
The specifications for tightening the bolts on a car engine's valve cover are given in terms of torque rather than in terms of how much force should be exerted on the tightening wrench. Why is torque...
-
The inversion temperature of hydrogen is (a) \(315 \mathrm{~K}\) (b) \(202 \mathrm{~K}\) (c) \(275 \mathrm{~K}\) (d) \(345 \mathrm{~K}\).
-
Dorsey Corporation purchased 90% of the common stock of Lansing Company on January 1, 2002. The cost of the investment was equal to the book value interest acquired. Lansing Company operates two...
-
If you had to speak in front of the Detroit City Council about this issue, who would you be supporting - the Hagermans or Judy Kelley? Explain your answer. People Are Making Big Money Kicking Detroit...
-
Richard chooses technique 0 and 2 requiring 10+10-20 efforts and provising 10+11=21 benefits. Hence, 21 is returned as the output Example 2: input1: 3 input2: (10,10,10,10) input3: (10,11,12,15)...
-
Predict the chemical shifts for the signals in the 1 H NMR spectrum of each of the following compounds: (a) (b) (c) (d) (e)
-
On January 2, 2007, a Sunny Communications $1,000 face value, six-year bond sold for $889. Investors who bought this particular bond will be paid interest equal to $40 every six months. Market...
-
Simplify the radical. 84
-
Conduct a coaching session Your coaching sessions need to include the following elements and is to be run in the following order: TASK 1 - Explanation: You are to explain what you will be...
-
What is an Instructional Coach (IC)? What is the purpose of the Instructional Coach? What are some of the roles of the Instructional Coach? What are the responsibilities of the Instructional Coach?...
-
Your basketball team has two coaches, Coach Mike and Coach Brian. Coach Mike decides the lineup for 60% of the games, and Coach Brian decides the lineup for 40% of the time. You love playing point...
-
A coaching company believes that coaching improves the SAT scores of high school students. To test their theory, they get two samples of students, one that was coached and one that was not. They...
-
Assuming Stock B is the market portfolio; Stock A Stock B Stock C Expected Return 0.032123 0.021519 0.011348 Variance 0.00175860 0.00114550 0 COV(AB) 0.00245250 If you believe the CAPM is valid, what...
-
The special products can be used to perform selected multiplications. On the left, we use (x + y)(x - y) = x 2 - y 2 . On the right, (x - y) 2 = x 2 - 2xy + y 2 . Use special products to evaluate...
-
Consider the discrete group G of order 8 that has the following Cayley diagram e If we have the sequence of operations: fcagec, which of the options represents the reduction of the sequence to a...
-
Consider 2-methylbutane (isopentane) Sighting along the C2C3 bond: (a) Draw a Newman projection of the most stable conformation. (b) Draw a Newman projection of the least stable conformation. (c)...
-
What are the relative energies of the three possible staggered conformations around the C2C3 bond in 2, 3-dimethylbutane? (See Problem 3.42)
-
Construct a qualitative potential-energy diagram for rotation about the CC bond of 1, 2-dibromoethane. Which conformation would you expect to be more stable? Label the anti and gauche conformations...
-
Provide an analysis of the benefits and challenges of having a growth mindset as opposed to having a fixed mindset. Examine the benefits of each type of mindset, as well as the challenges that each...
-
Discuss and explain what is Performance Management (P M)? How does P M fit into corporate strategy? What's in it for me? How does it work? What are my responsibilities? How does PM relate to other...
-
A conducting spherical shell has inner radius 2 / 3 R and outer radius R . ( a ) Suppose we put a charge Q onthe shell ( a conductor ) so that all of the charge goes to the outer radius. Find the...

Study smarter with the SolutionInn App


