Predict the major product expected for each of the following reactions: (a) (b) (c) (d) (e) (f)
Question:
(a)
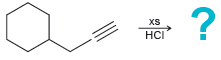
(b)
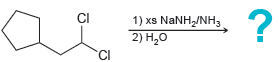
(c)
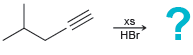
(d)
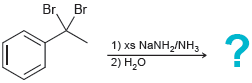
(e)
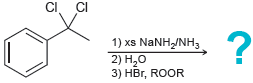
(f)
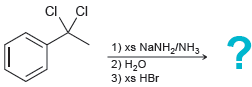
Transcribed Image Text:
? XS HCI CI 1) xs NANH/NH3 2) H,0 CI
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 45% (11 reviews)
a b ...View the full answer

Answered By

GERALD KAMAU
non-plagiarism work, timely work and A++ work
4.40+
6+ Reviews
11+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the major product from each of the following reactions. (a) (b) (c) (d) (e) (f) OH SOC, pyr OH HBr NaNH2 OH OH (1) TsCI, pyr (2) EtSNa Nal, H2SO OH
-
Predict the major product (or products) that would be obtained when each of the following compounds is nitrated: (a) (b) (c) OH CF CN SO3H OCH3 NO2
-
Predict the major product (or products) formed when each of the following reacts with a mixture of concentrated HNO3 and H2SO4. (a) (b) (c) 4-Chlorobenzoic acid (d) 3-Chlorobenzoic acid (e)...
-
You are attempting to arrive at what the monthly rate of change that has recently occurred in the real estate market. In doing some market research, you find two properties that have sold twice...
-
You wish to boil 1.1 kg of water, which has a specific heat capacity of 4186 J/kg-K. The water is initially at room temperature (293 K). Water boils at 373 K. 1) How much energy must be added to the...
-
Harley Company requires a minimum cash balance of $5,000. When the company expects a cash deficiency, it borrows the exact amount required on the first of the month. Expected excess cash is used to...
-
As winds blow past buildings, complex flow patterns can develop due to various factors such as flow separation and interactions between adjacent buildings. Assume that the local gage pressure, \(p\),...
-
Carson Corporation produces and sells three products, Alpha, Beta, and Gamma, in a local market and in a regional market. At the end of the first quarter of the current year, the following income...
-
Critically analyze the following question. PROVIDE REFERENCES What job factors are most important in the workplace of today?
-
Barfield Corporation prepares business plans and marketing analyses for start-up companies in the Cleveland area. Barfield has been very successful in recent years in providing effective service to a...
-
Describe sustainability and the triple bottom line. What is the difference between sustainability, corporate social responsibility, and being green?
-
Describe current wetland protection policies in the United States and give their strengths and weaknesses.
-
A firm can lease a truck for 4 years at a cost of $30,000 annually. It can instead buy a truck at a cost of $80,000, with annual maintenance expenses of $10,000. The truck will be sold at the end of...
-
Describe three types of systems testing and their importance for project success.
-
Identify the two descriptions below as being the result of either cost-push inflation or demand-pull inflation. a. Real GDP is below the full-employment level and prices have risen recently. b. Real...
-
Suppose that actual inflation is 3 percentage points, the Feds inflation target is 2 percentage points, and unemployment is 1 percent below the Feds unemployment target. According to the Taylor rule,...
-
Is it accurate to think of a fixed exchange rate as a simultaneous price ceiling and price floor? Explain.
-
List five key metrics that are useful to manage project progress.
-
Differentiate. y = 1/t 3 + 2t 2 - 1
-
6. (Potential Energy and Conservation of Energy) What should be the spring constant k of a spring designed to bring a 1200-kg car to rest from a speed of 95 km/h so that the occupants undergo a...
-
The reactions shown below are unlikely to occur as written. Tell what is wrong with each, and predict the actualproduct. OCCH3)3 . H2CH Br (a) CHCH-CH (CH]l3c (b) Na* - CI LCH3 LCH3 (c) Socil,...
-
Order each of the following sets of compounds with respect to SN1reactivity: H3C CH3 C NH2 (a) i CH3CH2CHCH3 CI CH (b) (CH)3CI (CH)Br (CH)3C (c) Br CHCH3 C-Br CBr 3
-
Order each of the following sets of compounds with respect to SN2reactivity: CI CH (a) CH3CH2CHCH3 H CH3CH2CH2CI CH (b) CH CH CH CHCCH2Br CHH CH2Br Br CH (c) CH3CH2CH20CH3 CHCH2CH2Br CHCH2CH20Tos
-
A Quality Management System, often called a QMS, is a set of internal rules that are defined by a collection of policies, processes, documented procedures, and records. This system defines how a...
-
1. Mr. Alvis, the director of Match Lighting, has found himself with sorts of emails including customer complaints about incorrect order deliveries, periodic reports on products, stock, and sales,...
-
A nursing team is having a routine meeting. One of the nurses, Stephen, is at the end of a 12-hour shift, and another nurse, Tanya, is just beginning hers. Tanya is a senior nurse in the unit with...

Study smarter with the SolutionInn App


