Predict the major product for each of the following intramolecular reactions: (a) (b) NH ? [H7 -H20
Question:
Predict the major product for each of the following intramolecular reactions:
(a)
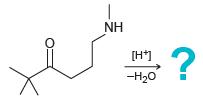
(b)
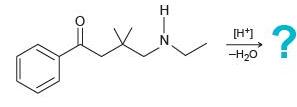
Transcribed Image Text:
NH ? [H7 -H20
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 42% (7 reviews)
a ...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the major product for each of the following reactions: a. b. c. d. e. f. g. h. i. j. CI :? NaSH DBN OTs
-
Predict the major product for each of the following transformations: (a)
-
Predict the major product for each proposed Diels-Alder reaction. Include stereochemistry where appropriate. (a) (b) (c) Ph 0 Ph
-
(a) Using the ion-pair equilibrium constant from Appendix J, with activity coefficients = 1, find the concentrations of species in 0.025 M MgSO 4 . Hydrolysis of the cation and anion near neutral pH...
-
Opulence Corporation operates several large cruise ships. One of these ships, the Bellwether, can hold up to 3,800 passengers and cost $760 million to build. Assume the following additional...
-
Explain why an organizations culture is important to an organization.
-
A game of chance based on a spinning wheel is available that pays \(n\) times money bet in the case of a win and nothing in the case of a loss. A gambler has developed a device by which he may...
-
Olympic Sports has two issues of debt outstanding. One is a 9% coupon bond with a face value of $20 million, a maturity of 10 years, and a yield to maturity of 10%. The coupons are paid annually. The...
-
Subject: operations management list and explain the items that go into and the items that come out of an aggregate plan and their importance 2 answers
-
A graduating student keeps applying for jobs until she gets an offer. The probability of getting an offer at any trial is 0.35. a. What is the expected number of applications? What is the variance?...
-
The data below were from the same 25-yearold participants described in How It Works 12.1, but now the scores represent the oldest age that would be acceptable in a dating partner. 25-year-old women...
-
Identify the reactants that you would use to make each of the following enamines: (a) (b) (c) -N
-
The following data refer to Clear Panes, a division of Global Corporation. Clear Panes makes and sells residential windows that sell for $150 each. Clear Panes expects sales of 150,000 units in 2017....
-
The moon is 3.8 x 10 km away frein the earth. How long a time will pass between the instant an astronaut on the moon speaks and the instant his veice is heard on earth? (His voice travels by laser...
-
A -6.80nC charge of mass 4.40E-6 kg is released from rest 4.10mm above a very large plane of positive charge. The charge accelerates toward the plane and collides with a speed of 4.50m/s. What is the...
-
As the bat flies at a constant speed of 0.04 v toward a large tree trunk (where v denotes the speed of sound) the bat emits an ultrasonic pulse. The pulse is reflected off the tree and returns to the...
-
When a car that has rolled down hill is stopped with the brakes, does energy move from the air to the car or the car to the air? Explain
-
2. If you were to go to the Moon to do the same measurement, in order to get the same answer for the density, what kind of scale (a beam scale or an electronic scale) you would bring with you to the...
-
A bean bag is randomly thrown onto the square table top shown below and does not touch a line. Determine the probability that the bean bag lands on An area that is not green.
-
Tanaka Company's cost and production data for two recent months included the following: March April Production (units).........300................600 Rent.....................$1,800............$1,800...
-
Trichloromethane (CHCl3, also called chloroform) has a larger dipole moment than CFCl3. Use three-dimensional structures and bond moments to explain this fact.
-
Indicate the direction of the important bond moments in each of the following compounds (neglect C-H bonds). You should also give the direction of the net dipole moment for the molecule. If there is...
-
Write structural formulas for all of the alkenes with (a) The formula C2H2Br2 and (b) The formula C2Br2Cl2. In each instance designate compounds that are cis-trans isomers of each other. Predict the...
-
Given the matrix A -3 0 -14 3 4 6 9115 7701 a) Determine all solutions of the homogeneous system Ax = 0. b) Determine if the columns of A span R.
-
Summarize each data source and include them for substance abuse and alcohol in the military. Analyze each data source for substance abuse and alcohol in the military for trustworthiness and accuracy....
-
On January 1, 2023, Holland Corporation paid $7 per share to a group of Zeeland Corporation shareholders to acquire 60,000 shares of Zeeland's outstanding voting stock, representing a 60 percent...

Study smarter with the SolutionInn App


