Predict the major product of each reaction below: (a) (b) (c) 1) EtMgBr 2) H20 :? H.
Question:
(a)
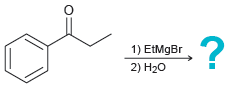
(b)
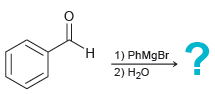
(c)
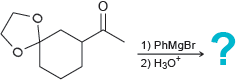
Transcribed Image Text:
1) EtMgBr 2) H20 :? H. 1) PhMgBr 2) H20
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
a...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the major product of each reaction below: (a) (b) (c) RCO3H RCO, ? H RCO3H
-
Predict the major product of the following reaction, and give a mechanism to support your prediction. NBS, h CH,CH ethylbenzene
-
Predict the major product for each reaction below: (a) (b) 1) [H'), HS 2) Raney Ni SH 1) [H*), HS' `H. 2) Raney Ni SH
-
Prove that there exist two languages A and B that are Turing-incomparablethat is, where A T B and B T A.
-
One of your corporate clients has approached you about whether or not its employees are required to include certain benefits provided by the corporation in their income. In particular, the...
-
A software company has designed a new version of its search engine, and wants to investigate consumer response. A randomly selected group of users of the old search engine is selected, and given...
-
A cloth manufacturer finds that 1 in every 400 shirts produced is faded. Find the probability that (a) the first faded shirt is the eighth item produced, (b) the first faded shirt is the first,...
-
A hotel housekeeper, Alison Harvey, was observed five time son each of four task elements, as shown in the following table. On the basis of these observations, find the standard time for the process....
-
After all of the account balances have been extended to the Balance Sheet columns of the work sheet, the totals of the Debit and Credit columns are $28,246 and $21,306, respectively. What is the...
-
Failure to follow accounting principles causes immense confusion, which in turn creates a number of problems for the organization. Those with vast experience in bookkeeping, however, are able to...
-
When 2 moles of benzaldehyde are treated with sodium hydroxide, a reaction occurs in which 1 mole of benzaldehyde is oxidized (giving benzoic acid) while the other mole of benzaldehyde is reduced...
-
Identify the reagents necessary to accomplish each of the transformations below: (a) (b) Me
-
Give a brief description of two emerging issues confronting the psychology profession. Then, explain how each issue might impact the future of training and practice in professional psychology.
-
In addition to managing expatriate employees, what other functions are important responsibilities for IHR to address?
-
What kind of training should companies be offering to recruit and develop local management talent, particularly in emerging markets?
-
One reason for expatriate assignment failurean employees premature return from an international assignmentis culture shock. What is culture shock? Provide some examples of culture shock contributing...
-
How does a global management team help a company to coordinate host country and cross-border business?
-
Many MNCs choose divergent labor practices, rather than convergent, and adapt their HR policies to local conditions. Find examples of companies that have been accused of running sweatshops and...
-
Convert the following hexadecimal numbers to decimal. Show your work. (a) A5 16 (b) 3B 16 (c) FFFF 16 (d) D0000000 16
-
I frequently use NY Times and CNN and am aware of Fox News but I never use it. I visit these sites, NY Times and CNN, a few times a week whenever I have to research something or see something on...
-
Malonic acid, HO2CCH2CO2H, is a diprotic acid. The pKa for the loss of the first proton is 2.83; the pKa for the loss of the second proton is 5.69. (a) Explain why malonic acid is a stronger acid...
-
At 25oC the enthalpy change, Ho, for the ionization of trichloroacetic acid is +6.3 kJ mol-1 and the entropy change, So, is +0.0084 kJ mol-1 K-1. What is the pKa of trichloroacetic acid?
-
Use the curved-arrow notation to write the reaction that would take place between dimethylamine (CH3)2NH and boron trifluoride. Identify the Lewis acid, Lewis base, nucleophile, and electrophile and...
-
In 2019, Rylan Enterprises' net income increased by $2.5 million while its depreciation expense decreased by $500,000, accounts receivable increased by $2,000,000 and accounts payable increased by...
-
Sweeten Company had no jobs in progress at the beginning of March and no beginning inventories. The company has two manufacturing departments-Molding and Fabrication. It started, completed, and sold...
-
The Sky Blue Corporation has the following adjusted trial balance at December 31. Cash Accounts Receivable Prepaid Insurance Notes Receivable (long-term) Equipment Accumulated Depreciation Accounts...

Study smarter with the SolutionInn App


