Predict the major product of the following reaction. HOS Dilute H2SO. ,5S. O,H
Question:
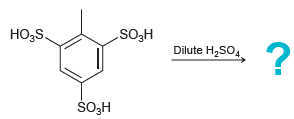
Transcribed Image Text:
H®OS Dilute H2SO. Но,5S. ŠO,H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
HOS S...View the full answer

Answered By

Ashok Kumar Malhotra
Chartered Accountant - Accounting and Management Accounting for 15 years.
QuickBooks Online - Certified ProAdvisor (Advance - QuickBooks Online for 3 years.
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the major product of each reaction below: (a) (b) (c) 1) EtMgBr 2) H20 :? H. 1) PhMgBr 2) H20
-
Predict the major product of each reaction below: (a) (b) (c) RCO3H RCO, ? H RCO3H
-
Predict the major product for each E1 reaction: a. b. eat Br H2SO4 eat
-
Perform the indicated operations. Let k be a natural number. a. (-2) c. (-2)4 e. Is (-2)2+ positive or negative? b. (-2) d. (-2)5
-
How can an entrepreneur seeking funds to launch a business convince potential lenders and investors that a market for the product or service really does exist?
-
Refer to the Arctic Springs Filtration Department Data Set. Complete Step 5 of the process costing procedure: Assign costs to units completed and to units in ending inventory. Prepare a schedule that...
-
Explain why a greedy method provides suboptimal solutions to the 0-1 knapsack problem but provides optimal solutions to the fractional knapsack problem. Could there be a search tree for which greedy...
-
For each of the following situations, identify the correct factor to use from Tables 1 or 2 in the appendix on present value tables. Also, compute the appropriate present value. 1. Annual net cash...
-
Hull Company reported the following income statement information for the current year: Sales $ 415,000 Cost of goods sold: Beginning inventory $ 139,500 Cost of goods purchased 278,000 Cost of goods...
-
Verify that (a) (2 - i) - i(1 - 2i) = -2i; (b) (2, - 3)(-2, 1) = (-1, 8) (c) (3, 1)(3-1)( TO 5 10 .
-
It is found that K P is independent of T for a particular chemical reaction. What does this tell you about the reaction?
-
The following transformations cannot be accomplished, even with the help of blocking groups. In each case, explain why a blocking group will not help. (a) (b) . NO2 Br
-
Toyota Motor Companys advertisement in The Economist magazine is entitled Your Toyota is My Toyota. It shows a photograph of a female production team member on the assembly line at the companys...
-
what is the difference between the types and frequency of reports between financial and managerial accounting?
-
Indicate whether the following statement is true or false and explain why Managerial accounting reports must follow generally accepted accounting principles and are audited by chartered professional...
-
Indicate whether the following statement is true or false and explain why Managerial accountants explain and report manufacturing and non-manufacturing costs and determine cost behaviours, but are...
-
Magic Mountain retires its 9% bonds for $128,000 before their scheduled maturity. At the time, the bonds have a face value of 126,000 and a carrying value of $117,000. Record the early retirement of...
-
Barnes Corp. issues $ 8 0 0 , 0 0 0 of 9 % , 2 0 - year bonds. The bonds are dated January 1 , 2 0 1 1 , and are due in 2 0 years on January 1 , 2 0 3 1 . Interest is paid semi - annually each June 3...
-
Find the area of the surface. The part of the sphere x 2 + y 2 + z 2 = 4 that lies above the plane z = 1
-
A consultant is beginning work on three projects. The expected profits from these projects are $50,000, $72,000, and $40,000. The associated standard deviations are $10,000, $12,000, and $9,000....
-
The physical basis of some carbon monoxide detectors is the infrared detection of the unique stretching vibration of carbon monoxide at 2143 cm-1. How many times per second does this stretching...
-
The ==C-H stretching absorption of 2-methyl- I-pentene is observed at 3090 cm-1. If the hydrogen were replaced by deuterium, at what wavenumber would the ==C-D stretching absorption be observed?...
-
The ==C-H stretching absorption of 2-methyl- I-pentene is observed at 3090 cm-1. If the hydrogen were replaced by deuterium, at what wavenumber would the ==C-D stretching absorption be observed?...
-
Speculate sales : Speculate on the projected sales. Justify your product or service by the numbers; discuss your projected revenue gain. Note: it MUST have an ROI that justifies the project for...
-
8.6 In Figure P8.6 the experimental unit step response of a process system is shown. (a) What is the transfer function that can be used to best describe the system dynamics among the following. 1 (1)...
-
An 84 year old female arrives to the ER today with her son with a complaint of new onset of confusion. What are your differential diagnoses for a patient with new altered mental status? Go through...

Study smarter with the SolutionInn App


