Predict the major product(s) for each of the following reactions: 1) MCPBA 1) , THF 2),, NaOH
Question:
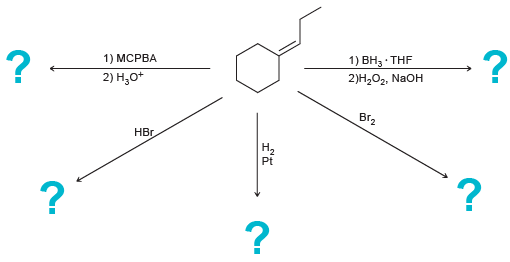
Transcribed Image Text:
1) MCPBA 1) ВН, THF 2)Н,О, NaOH 2) Н,о* ВГz НЕ. На Pt
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (17 reviews)
HO OH En Br 1 M...View the full answer

Answered By

Hamza Amjad
Currently I am student in master degree program.from last two year I am tutring in Academy and I tought many O/A level student in home tution.
4.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the major product (or products) formed when each of the following reacts with a mixture of concentrated HNO3 and H2SO4. (a) (b) (c) 4-Chlorobenzoic acid (d) 3-Chlorobenzoic acid (e)...
-
Predict the major product from each of the following reactions. (a) (b) (c) (d) (e) (f) (g) (h) (i) CH NH2, cat. HA N-H, cat. HA NH2 cat. HA PPha (1) HS SH (2) Raney Ni, H2 0 CH2PPha (excess) O. O...
-
Predict the major product from each of the following reactions. (a) (b) (c) (d) (e) HO OH NH2 HA (cat.) (1) HCN (2) LiAIH4 (3) H2o mCPBA
-
Assume that 3-month Treasury bills totaling $23 billion were sold in $10,000 denominations at a discount rate of 5.200%. In addition, the Treasury Department sold 6-month bills totaling $21 billion...
-
Develop and prototype a new interface design for the system's function using Graphical User Interface. Include common interface functions such as (textboxes, radio button, drop-down menus, check...
-
Carrboro has three equal-sized groups of people: (1) type A people consistently prefer more police protection to less; (2) type B people prefer high levels of police protection to low levels and they...
-
Consider ethyl-tertiary-butyl-ether (ETBE) as an alternative gasoline oxygenate to MTBE. Although the latter appears to have the best combination of properties such as oxygen content, octane number,...
-
Michael and Mary Mason sold for $380,000 in November of 2012 their residence that they had purchased in 2002 for $75,000. They made major capital improvements during their 10-year ownership totaling...
-
a. Draw a flowchart of a program called CalculateAverage that asks the user to enter 3 integers (using a loop), then calculates and displays the average of the numbers. b. Trace the execution of the...
-
The following arrangement indicates whether 60 consecutive cars that went by the toll booth of a bridge had local plates, L, or out- of- state plates, O: L L O L L L L O O L L L L O L O O L L L L O L...
-
Would water be a suitable proton source to protonate the following compound? ONa
-
When 3-bromo-3-ethylpentane is treated with sodium acetylide, the major products are 3-ethyl-2-pentene and acetylene. Explain why the carbon skeleton does not change in this case, and justify the...
-
In a recent, USA Today article titled Consumers Are Caught in a Sugar Battle, Americans consume an average of 22.2 teaspoons of sugar per day. Assume daily sugar consumption follows the normal...
-
A cereal company runs a certain advertisement in three media: newspaper, radio, and TV. Of the customers surveyed, 40 saw the advertisement on TV, 40 heard it on the radio, 30 read it in the...
-
What insights can be found from a comparison of Haiti and the DR for the potential role of environment in economic development?
-
Suppose that a study finds that there is complementarity in fertility decisions. What would this mean? What are the possible implications?
-
What are the strengths and weaknesses of policy options for nations whose populations begin declining before they become a high-income country?
-
Here are some questions to review for the Progresa/Oportunidades Programme, examined in the end-of-chapter case study: a. What is the Progresa/Oportunidades Programme, and what does it try to...
-
Sketch a graph of a function that is continuous on (-, ) and has the following properties. Use a sign graph to summarize information about the function. f(0) = f(4) = f'(0) = f'(2) = f'(4) = 0; f(x) ...
-
As indicated by mutual fund flows, investors tend to beat the market seek safety invest in last year's winner invest in last years loser
-
Explain why the reaction of the cis-isomer of this compound with potassium tert-but oxide in tert-butanol is about 500 times faster than that of thetrans-isomer. Br C(CH3)3
-
Explain which of these compounds has a faster rate of E2elimination: CH CH3 CI 'CI
-
Frequently, several different routes can potentially be used to synthesize a desired compound. For example, the following two routes can be envisioned for the preparation of cyclopentyl methyl ether....
-
A 9 Loan amount Annual interest rate Number of years until maturity Annual payment amount B $300,000.00 Date Graded Worksheet 7% 4 On January 1, 2024, a company borrows cash from a local bank by...
-
As we have seen in this course, virtually every generation has felt that the existing threat to its privacy was unprecedented in nature. That said, the sheer capacity to collect metadata which exists...
-
In 1982 the inflation rate hit 16%. Suppose that the average cost of a textbook in 1982 was $20. What was the expected cost in the year 2017 if we project this rate of inflation on the cost? (Assume...

Study smarter with the SolutionInn App


