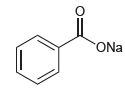
Would water be a suitable proton source to protonate the following compound? ONa
Question:
Transcribed Image Text:
ONa
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 45% (11 reviews)
No water would not be a suitable proto...View the full answer

Answered By

HABIBULLAH HABIBULLAH
I have been tutor on chegg for approx 5 months and had solved a lot of questions.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
We will learn all of the following reactions in upcoming chapters. For each of these reactions, notice that the product is an anion (ignore the positively charged ion in each case). In order to...
-
Would ethanol (CH 3 CH 2 OH) be a suitable solvent in which to perform the following proton transfer? Explain your answer: ONH2 + NH3 NH2 H.
-
The following compound is known to be chiral. Draw its enantiomer, and explain the source of chirality. CH
-
Prepare a company analysis (strengths and weaknesses) Instructions - Prepare the SW portion of a SWOT analysis as a report. Use your textbook and the instruction below. Prioritizing the Results: The...
-
Investigate learning management software packages that are available through application service providers. Using the World Wide Web, identify at least two potential sources of such software. What...
-
Every year, the World Bank rates countries on the basis of their quality of governance, along a number of different dimensions (such as political stability, government effectiveness, and the rule of...
-
For the ammonia process in Example 6.4, consider operation of the reactor at \(932^{\circ} \mathrm{F}\) and \(400 \mathrm{~atm}\). Use a simulator to show how the product, recycle, and purge flow...
-
Here is the income statement for Eberle, Inc. Additional information: 1. Common stock outstanding January 1, 2014, was 32,000 shares, and 40,000 shares were outstanding at December 31, 2014. 2. The...
-
In a cache and memory system with the following characteristics: Direct Mapped 16 KByte 32 Byte Cache Lines (Blocks) 32 bit addresses, byte addressable How many bits are the Tag, Index and Offset...
-
ABC Framing has been hired to frame a light commercial building. The project began on July 2 and was completed on August 9. The following is a list of accounting transactions associated with the...
-
There are four constitutional isomers with molecular formula C 3 H 9 N. Draw a Lewis structure for each isomer and determine the number of lone pairs on the nitrogen atom in each case?
-
Predict the major product(s) for each of the following reactions: 1) MCPBA 1) , THF 2),, NaOH 2) ,* z . Pt
-
1. How does McCain view her role as human resource manager? 2. What is Harrisons business strategy? 3. What is the structure and staffing of HR? 4. Analyze the data in Exhibit 1.4. What are its...
-
What aspects of population policy alternatives including their strengths and weaknessesare illustrated by the cases of China and India?
-
Explain the differences between characteristics of public goods and private goods and services. How can goods and services provided by nongovernmental organisations be considered in this framework?
-
If a reform improves everyones income on average, why might people vote against it? You may wish to provide one or more numerical examples to illustrate your answer.
-
What can be learned from the historical experiences of Costa Rica and Guatemala about internal conflicts, and about both positive and negative foreign influences?
-
What lessons can be learned for low-income countries from Botswanas successes?
-
Locate the critical points of the following functions. Then use the Second Derivative Test to determine (if possible) whether they correspond to local maxima or local minima. f(x) = 2x 2 ln x - 11x 2
-
4. Jobe dy -Y 2 et by
-
Show the products of this reaction. How would the composition of the products change if t-BuO ? in t-BuOH were used in place of ethoxide ion in ethanol? ELOH + CH,CH,0
-
All of the stereo isomers of 1, 2, 3, 4, 5, 6-hexachlorocyclohexane have very similar rates of E2 reaction except the following stereo isomer, which reacts about 7000 times more slowly than the...
-
Show the product of thisreaction: Ph Br- - + NaOEt Br EIOH Ph
-
es Hart, Attorney at Law, experienced the following transactions in Year 1, the first year of operations: 1. Accepted $16,600 on April 1, Year 1, as a retainer for services to be performed evenly...
-
Dahlia Corporation has a current accounts receivable balance of $439,516. Credit sales for the year just ended were $5,503,810. a. What is the receivables turnover? Note: Do not round intermediate...
-
Why does the organizational structure hold political significance? Provide an in-depth analysis of this concept using examples from both academic literature and real-world instances. Additionally,...

Study smarter with the SolutionInn App


