Predict the product and draw the mechanism for each of the following reactions: a. b. c. 1)
Question:
a.
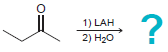
b.
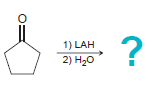
c.
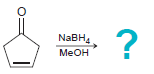
Transcribed Image Text:
1) LAH 2) H20 1) LAH 2) H20
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (6 reviews)
a b c ...View the full answer

Answered By

ABHISHEK S
I started teaching when I was studying at college .I taught some of my juniors in college.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each of the following reactions, predict the product and draw the mechanism of its formation. a. b. c. d. e. f. 1) PhMgBr 2) H20 Me 1) NaCN 2) H20 *Me
-
For each reaction, predict the product and draw the mechanism of its formation. a. b.
-
Draw a mechanism for each of the following E1 processes: a. b. c. d. H,SO, Heat Br ETOH, Heat
-
If you can't find similar ratios to class-covered ones, use financial formulas to calculate them manually. Choose up to two of the following: Operating Margin EBITDA Margin Payout Ratio 3. Analysis...
-
Selected data from Komar Company follow. a. Compute the accounts receivable turnover for 2014. (Round your answer to 2 decimal places Accounts receivable turnover: times b. Compute the inventory...
-
In the case of Pampers, why is it important to establish a relationship with the buyers of its products and their families?
-
How is art used in support of social structure and power?
-
On January 1, 2011, TCU Utilities issued $1,000,000 in bonds that mature in 10 years. The bonds have a stated interest rate of 10 percent and pay interest on June 30 and December 31 each year. When...
-
8. Construct a minimize Deterministic Finite Automata for the following Finite Automata (using Myhill Nerode method) 1 0 0 A B 0 0 0,1 1 0 H 1
-
On February 11, 20Y9, Quick Fix Company purchased $2,250 of supplies on account. In Quick Fixs chart of accounts, the supplies account is No. 15, and the accounts payable account is No. 21. a....
-
A bottle at 325 K contains an ideal gas at a pressure of 162.5 10 3 Pa. The rubber stopper closing the bottle is removed. The gas expands adiabatically against P external = 120.0 10 3 Pa, and some...
-
Predict the product and draw the mechanism for each of the following reactions: a. b. c. ? 1) LAH 2) H20
-
What are the issues facing an entrepreneur in deciding whether or not the business needs to be put into bankruptcy today?
-
List four ways that related claims are handled to save costs and time, and their pertinent Federal Rules of Civil Procedure. Compare in rem and quasi in rem actions. How may a plaintiff respond to...
-
Each province in Canada has passed legislation that governs the local sale of goods transactions. There is no Federal sale of goods transaction act in Canada. Do you believe that a Federal sale of...
-
Why does Jeff say he only drinks socially and does not have a drinking problem? How would you respond to this and his contention that he is only in treatment to satisfy his wife and lawyer?
-
What is the right to education in international law and its importance? Explain
-
How do moral obligations apply to business organizations? What are the major arguments concerning corporate responsibility.?
-
Two buildings in a sports complex are shaped and positioned like a portion of the branches of the hyperbola 400x 2 - 625y 2 = 250,000, where x and y are in meters. (a) How far apart are the buildings...
-
At Glass Company, materials are added at the beginning of the process and conversion costs are added uniformly. Work in process, beginning: Number of units Transferred - in costs Direct materials...
-
Propose a structure for an octapeptide that shows the composition Asp, Gly2 Leu, Phe, Pro2, Val on amino acid analysis. Edman analysis shows a glycine N-terminal group, and leucine is the C-terminal...
-
The reaction of ninhydrin with an ?-amino acid occurs in several steps. (a) The first step is formation of an imine by reaction of the amino acid with ninhydrin. Show its structure and the mechanism...
-
Draw resonance forms for the purple anion obtained by reaction of ninhydrin with an a-amino acid.
-
How do the resource descriptions (typing) used in the Incident Command System lead to a more effective response?
-
A company incurs $4172000 of overhead each year across three departments: Ordering and Receiving, Mixing, and Testing. The company prepares 2000 purchase orders, works 50000 mixing hours, and...
-
Hemming Company reported the following current-year purchases and sales for its only product. Date January 1 January 10 Activities Beginning inventory March 14 March 15 July 30 Sales Purchase Sales...

Study smarter with the SolutionInn App


