Propose a mechanism that explains formation of the following product: H2SO4 Heat
Question:
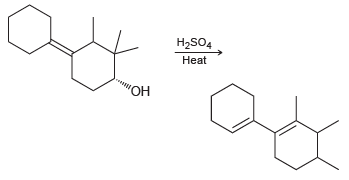
Transcribed Image Text:
H2SO4 Heat "ОН
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 23% (13 reviews)
OH sofor ...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Write a mechanism that explains formation of the products shown in the following reaction. Br, NaCI, H20o Rr CL
-
Provide a mechanism that explains formation of the following products. Include all intermediates, formal charges, and arrows showing electron flow. 0 N-Br (NBS) 0 hv 11
-
Provide a mechanism that explains formation of the following products. CI HCI (concdCI+
-
Access various employment Web sites (for example, www.monster.com and www.dice.com ) and find several job descriptions for a database administrator. Are the job descriptions similar? What are the...
-
Write a paper, response to the following question. Do not use the question in your response. Read the case study on Undercover Advertising "Viral Marketing." After reading the case study, take a...
-
The following events occurred during the month of September 2024 for the business of Shrutis Webdesign Service. Required (a) Determine the effects of business transactions on a statement of financial...
-
Solve Problem 11.6 by changing \(c\) to 2 . Data From Problem 11.6:- Find the free-vibration response of a viscously damped single-degree-of-freedom system with \(m=k=c=1\), using the central...
-
Rise N Shine Coffee Company produces Columbian coffee in batches of 6,000 pounds. The standard quantity of materials required in the process is 6,000 pounds, which cost $ 5.50 per pound. Columbian...
-
In Texas, on his annual renewal form, an RN stated that he got a second DWI 9 months ago. What do you tell him will be the most likely decision as to his license? If you are not in the state of...
-
The manufacturing plant of National Brand Corporation has agreed to supply its best 2 customers with their number 1 product. Customer A will be supplied for 3 months and customer B will be supplied...
-
Draw all constitutional isomers of C 4 H 9 Br, and then arrange them in order of increasing reactivity toward an E2 reaction.
-
(S)-1-Bromo-1,2-diphenylethane reacts with a strong base to produce cis-stilbene and trans-stilbene: a) This reaction is stereo-selective, and the major product is trans-stilbene. Explain why the...
-
Fairfield Services Company, from Problem 4-3A, prepared an end-of-period spread-sheet (work sheet) a part of which is shown below, at December 31, 2015, the end of the current fiscal year (shown...
-
Basic Partnership, a cash-basis calendar-year entity, began business on February 1, 2007. Basic incurred and paid the following in 2007: Filing fees incident to the creation of the partnership $...
-
How many types of basic types of inventory control systems are there?
-
There is a negative association between the supply of affordable housing and homeless rates in cities. If this hypothesis is correct, what change would you predict would occur in homeless rates in...
-
There is a positive association between the percentage of the civilian labor population unemployed in a state and the number of firearm deaths in that state. Based on this hypothesis, what change...
-
What challenges do economies face in light of optimal allocation and public goods? Explain in detail.
-
Evaluate each sum as described. The sum of the first 80 positive integers
-
The population of Detroit, Michigan, decreased from 1,027,974 in 1990 to 688,701 in 2013 (Source: U.S. Census Bureau). Find the average rate of change in the population of Detroit, Michigan, over the...
-
Predict the number of carbon resonance lines you would expect in the 13C NMR spectra of the following compounds: (a) Methylcyclopentane (b) 1-Methylcyclohexene (c) 1, 2-Dimethylbenzene (d)...
-
Propose structures for compounds that fit the following descriptions: (a) A hydrocarbon with seven lines in its 13C NMR spectrum (b) A six-carbon compound with only five lines in its 13C NMR spectrum...
-
Assign the resonances in the 13C NMR spectrum of methyl propanoate, CH3CH2CO2CH3(figure). TMS CH-cH 2 1 120 200 180 160 140 100 20 40 O ppm 60 Chemical shift (8) Intensity
-
Assume we are using MergeSort on comparable Students who are compared with respect to their gpa. Assume we have an array of 8 students: [{name:"Ali", gpa:2.9}, {name:"Aye", gpa:3.1}, {name:"Sevil",...
-
Consider a two-input neuron that multiplies its two inputs x1 and x2 to obtain the output 'o'. Let L be the loss function that is computed at 'o'. Suppose that if the values of 044 = 5, x1 = 2, and...
-
The classification problem with four classes of input vector are Class 1: {p = [1], P2 = [2]}, Class 2: {p3 = [21],P4 = []}, Class 3: {ps = [2], P6 = []}, Class 4: {p7 = [1], P = [2]} Design a...

Study smarter with the SolutionInn App


