Propose a plausible synthesis for each of the following transformations: a. b. c. d. e. f. g.
Question:
a.
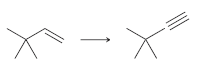
b.
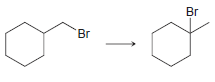
c.
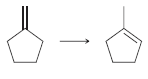
d.
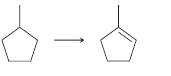
e.
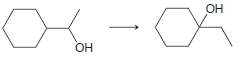
f.
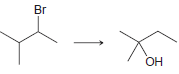
g.
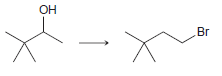
h.
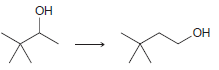
Transcribed Image Text:
Br Br
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (8 reviews)
a b c d ...View the full answer

Answered By

Hamza Amjad
Currently I am student in master degree program.from last two year I am tutring in Academy and I tought many O/A level student in home tution.
4.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Propose a plausible synthesis for each of the following transformations: (a) (b) (c) (d) (e) (f) Br Br
-
Propose a plausible synthesis for each of the following transformations: (a) (b) (c) (d)
-
Propose a plausible synthesis for each transformation. a. b. c. d. e. f. g. h. i. j. k. l. m. n. o. p. q. r. s. t. u.
-
The regression of the X and Y equation is Y = 48.32 -2.105X, What is the slope of the data.
-
1. How would you apply the concepts of identity, power, and advocacy when working with clients of a different gender, race, ethnicity, social class, or sexual orientation? Why do you think these...
-
A currently owned shredder originally costing \($800,000\) was purchased 6 years ago for use in a refuse-powered electrical generating plant. It was depreciated as MACRS-GDS 5-year property due to...
-
Why would a company want to become sustainable?
-
Specialized Consulting Service Company's after-tax net cash flows associated with two mutually exclusive projects, Alpha and Beta, are as follows: a. Calculate the net present value for each project...
-
Function f is given by f(x) = 2.5x + 3x3 - 2.6x - 5.1x - 5.6 Part A doniuslso a luodite collige ovito in aldsollqga roda noisoup a lo haq dass so On the closed interval [-.05, 1.2], determine the...
-
Find a recent engineering failure in the news and prepare a technical report of at least 250 words explaining how a mechanical engineer could have prevented the failure. Explain clearly how your...
-
Identify the reagents you would use to accomplish each of the following transformations: a. b.
-
Identify the reagents you would use to convert 2-bromo-2-methylbutane into 3-methyl-1-butyne.
-
Write Haworth formulas corresponding to the furanose forms of each of the following carbohydrates: (a) D-Xylose (c) L-Arabinose (b) D-Arabinose (d) D-Threose
-
Jeff purchased a card for $180 that gives him 20 visits to a new gym and includes a one time fee for unlimited use of the sauna. After 5 visits, he has $123.75 left on the card, and after 11 visits,...
-
A study examined transformer voltage sags and swells. For a sample of 106 transformers built for heavy industry, the mean number of sags per week was 40 and the mean number of swells per week was...
-
The temperature of a 50 g sample of aluminum is raised from 20C to 60C when 440 cal of heat are added. The specific heat capacity of the aluminum will be?
-
Provide a minimum of a 3-year history of the following: unemployment rate in the United States, inflation rate in the United States, and labor force participation rate in the United States. Indicate...
-
The following graph shows the market for euros, which is initially in equilibrium. Suppose an economic expansion in Canada leads to an increase in the incomes of Canadian households, causing imports...
-
Determine how many terms of the following convergent series must be summed to be sure that the remainder is less than 10 -4 in magnitude. Although you do not need it, the exact value of the series is...
-
Complete the equations for the following equilibria and calculate Keq where the Keq expression includes [HO]. Be sure to enter Keq in proper scientific notation. (a) ammonia (acting as a base) reacts...
-
The reaction of an alkyl chloride (or bromide) with sodium iodide in acetone proceeds according to the following equation: Sodium iodide is soluble in acetone, whereas both sodium chloride and sodium...
-
The Lucas test is used to check for the presence of an alcohol functional group in an unknown compound. The test reaction is shown in the following equation: Smaller alcohols are soluble in the...
-
Explain why this secondary alcohol reacts with HCl and ZnCl2 in H2O at about the same rate as a primary alcohol (see Problem 8.38) OH Cl-CH,CHCH3
-
A European call option for a share costs $5.00. The exercise price of the call option is $100.00. An investor buys one call and holds it until maturity: a. Under what circumstances will the holder of...
-
You have been asked to estimate the cost of capital for the UTX corporation. The company has 7 million shares and 150,000 bonds outstanding at par value $10,000. In addition, it has $300 million in...
-
MM Corp. has 50,000 shares outstanding with share price of $18. It has debt with market value of $300,000. The equity beta is 1.2 and debt beta is 0.1. The risk-free rate is 2% and the market risk...

Study smarter with the SolutionInn App


