Show two different methods for preparing each of the following alkenes (one method using a sterically hindered
Question:
a.
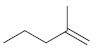
b.
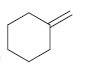
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (15 reviews)
a...View the full answer

Answered By

Munibah Munir
I've done MS specialization in finance’s have command on accounting and financial management. Forecasting and Financial Statement Analysis is basic field of my specialization. On many firms I have done real base projects in financial management field special forecasting. I have served more than 500 Clients for more than 800 business projects, and I have got a very high repute in providing highly professional and quality services.I have capability of performing extra-ordinarily well in limited time and at reasonable fee. My clients are guaranteed full satisfaction and I make things easy for them. I am capable of handling complex issues in the mentioned areas and never let my clients down.
4.60+
467+ Reviews
648+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Show at least two different methods for preparing 1-methylcyclohexene from 1-methylcyclohexanol.
-
Suggest appropriate methods for preparing each of the following compounds from the starting material of your choice. (a) CH3CH2CH2CH2CH2MgI (c) CH3CH2CH2CH2CH2Li (b) CH3CH2CCMgI (d)...
-
Suggest appropriate methods for preparing each of the following compounds from the starting material of your choice. (a) CH3CH2CH2CH2CH2MgI (b) CH3CH2 CPCMgI (c) CH3CH2CH2CH2CH2Li (d)...
-
What is the output of the following program: #include using namespace std; void Push(int x[], int y[], int n) { int i = 0, j = n - 1; while (i
-
A water-content test was made on a sample of clayey silt. The weight of the wet soil plus container was 18.46 g, and the weight of the dry soil plus container was 15.03 g. Weight of the empty...
-
Harmandeep Ltd. is a private company in the pharmaceutical industry. It has been preparing its financial statements in accordance with ASPE. Since it has plans to go public in the next 3 to 5 years,...
-
For what flowrate through the Venturi meter of Problem 3.127 will cavitation begin if \(p_{1}=275 \mathrm{kPa}\) gage, atmospheric pressure is \(101 \mathrm{kPa}\) (abs), and the vapor pressure is...
-
The unadjusted trial balance of Bens Jewelers on December 31, 2016, the end of its fiscal year, appears on shown below. INSTRUCTIONS 1. Copy the unadjusted trial balance onto a worksheet and complete...
-
Identify key performance indicators and outcome measures for a Shortage of nurses in healthcare. Do you believe people are born with the ability to persuade or influence others, or can people learn...
-
You have recently been appointed as the partner for the 2011 financial year audit of AllNatural (Pty) Ltd, a new client for your firm. AllNaturals financial year end is 30 September. As a result of a...
-
What are some of the long-term effects of pesticides on human health?
-
Identify the major and minor products for the E2 reaction that occurs when each of the following substrates is treated with a strong base: a. b. c. d. e. f. g. h. Br
-
(1) Define the term internal rate of return (IRR). What is each franchises IRR? (2) How is the IRR on a project related to the YTM on a bond? (3) What is the logic behind the IRR method? According to...
-
Tina walks into Teds sporting goods store and buys a punching bag for $100. That $100 payment counts as for Tina and for Ted. a. income; expenditure b. value added; multiple counting c. expenditure;...
-
Ralph Sample had the following income and deductions listed on his 2019 income tax return: Compute the amount of Ralphs 2019 net operating loss. Salary Business income Interest income on personal...
-
In a balanced system, the phasor sum of the line-to-line voltages and the phasor sum of the line-to-neutral voltages are always equal to zero. (a) False (b) True
-
How do free products make the calculation of GDP more difficult? What are hedonic adjustments and why are they necessary? Will inflation tend to be overstated or understated if quality improvements...
-
A balanced \(\Delta\)-load can be converted to an equivalent balanced- \(Y\) load by dividing the \(\Delta\)-load impedance by (a) \(\sqrt{3}\) (b) 3 (c) \(1 / 3\)
-
Calculate y'. y = (x 2 + x 3 ) 4
-
Consider model (9.18). What is the effect on the model parameter estimates, their standard errors, and the goodness-of-fit statistics when (a) The times at risk are doubled, but the numbers of deaths...
-
Using any alkyne needed, how would you prepare the following alkenes? (a) Trans-2-Octenc (b) Cis-3-Heptcne (c) 3-Methyl-1-pentene
-
The pK a of acetone, CH 3 COCH 3 , is 19.3. Which of the following bases is strong enough to de-protonate acetone? (a) KOH (p K a of H 2 O = 15.7) (b) Na + C CH (p K a of C 2 H 2 = 25) (c) NaHCO 3...
-
Show the terminal alkyne and alkyl halide from which the following products can he obtained. If two routes look feasible, list both. a) CHCH2CH2CEH (b) (CH/2HCH-CH3 (c)
-
Johnny's Lunches is considering purchasing a new, energy-efficient grill. The grill will cost $31,000 and will be depreciated straight-line over 10 years to a salvage value of zero. The grill will...
-
John wants to "roll in" or finance the loan fee of $3,800 into the loan amount which would make the loan $90,800 and the interest rate is 6%. Assume that the lender agrees to allow the loan fees to...
-
A company is faced with the decision of whether to lease or purchase new equipment. The equipment can be leased at an annual cost of $210,000 or purchased for $560,000. The equipment has an expected...

Study smarter with the SolutionInn App


