Show what reagents you would use to prepare each of the following ethers via a Williamson ether
Question:
a.
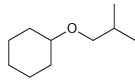
b.
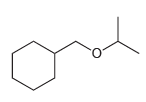
c.
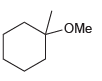
Transcribed Image Text:
OMe
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 90% (10 reviews)
a A Williamson ether synthesis will be more efficient with ...View the full answer

Answered By

Amar Kumar Behera
I am an expert in science and technology. I provide dedicated guidance and help in understanding key concepts in various fields such as mechanical engineering, industrial engineering, electronics, computer science, physics and maths. I will help you clarify your doubts and explain ideas and concepts that are otherwise difficult to follow. I also provide proof reading services. I hold a number of degrees in engineering from top 10 universities of the US and Europe.
My experience spans 20 years in academia and industry. I have worked for top blue chip companies.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Question 20 The relationship between services and providers is much stronger than that between products and producers. True False
-
What reagents would you use to prepare each of the following thiols: a. b. c. SH SH
-
Show the reagents you would use to achieve the following transformation: N. H.
-
Estimate the moment of inertia of a bicycle wheel.
-
1. Prepare a performance report showing both month and year-to-date data for Post Manufacturing's Machining Department for February, 2011 using the following data: 2. The following data was taken...
-
Gelinas Computer Company was organized on May 1. On that date, the company purchased 22,000 USB flash drives to be sold with personal computers, each pre-loaded with the companys product information...
-
The frictionless converging stationary nozzle of the hydraulic turbine shown in Fig. P12.46 has an inlet pressure \(p_{0}=480 \mathrm{kPa}\), a negligible inlet velocity \(V_{0}\), and an exit...
-
Harold H. Heidingsfelder signed a credit agreement as vice president of J. O. H. Construction Co. for a line of credit with Pelican Plumbing Co. The credit agreement contained the following language:...
-
Consider the following FIRE-INSURANCE PROBLEM where fire partially destroys a $500 house. EVENT FIRE PROBABILITY 0.02 OUTCOME $100 NO FIRE 0.98 $500 INSURANCE PAYOUT PREMIUM $400 0 ?? ?? (a) What do...
-
A ride hailing company has their DB structured in 3 major tables as described in the SCHEMA section below. Write a query to fetch the top 100 users who traveled the most distance using the service....
-
Consider the oxidation of the amino acid glycine NH 2 CH 2 COOH to produce water, carbon dioxide, and urea NH 2 CONH 2 : NH 2 CH 2 COOH(s) + 3O 2 (g) NH 2 CONH 2 (s) + 3CO 2 (g) + 3H 2 O(l)...
-
Calculate the average CH bond enthalpy in methane using the data tables. Calculate the percent error in equating the average CH bond energy in Table 4.3 with the bond enthalpy. Table 4.3 Selected...
-
Bottled gas for recreational vehicles contains propane, C 3 H 8 , and butane, C 4 H 10 . Which molecules have the faster velocity?
-
An article on bloomberg.com stated that not all currentaccount deficits are bad . . . [but] one problem with persistent deficits is that foreign investors can end up owning a big share of assets,...
-
Request the participation of managers from two companies and then ask them to fill out an entrepreneurial management scale. Based on the scale, which firm is more entrepreneurially managed? Does this...
-
Find three examples of firms that pioneered a new product in a new market and were able to achieve long-run success based on that entry. Find three examples of firms that were not the pioneers but...
-
Do you think the slope of the short-run aggregate supply (SRAS) curve will affect the size of the fiscal multiplier or not? Briefly explain.
-
Suppose a fellow student in your class makes the following comment: Budget deficits are always harmful to the economy and the government should never allow itself to spend more than what it receives...
-
Simplify the following Boolean equations using Boolean theorems. Check for correctness using a truth table or K-map. (a) (b) (c) Y = AC+ABC Y = AB+ABC+(A+T)
-
What are the 5 Cs of marketing channel structure?
-
Propose a mechanism to account for cleavage of the -cyanoethyl protecting group from the phosphate groups on treatment with aqueous ammonia. (Acrylonitrile, H 2 C = CHCN, is a by product.) What kind...
-
Identify the following bases, and tell whether each is found in DNA, RNA, or both: (c) (b) (a)
-
Identify the following nucleotide, and tell how it is used:
-
How is decision-making in organizations best supported by Management Accounting (a descriptive nature) and how should it be supported by Management Accounting (a prescriptive nature)? Is there a gap...
-
Discuss the thermodynamic principles governing the separation of azeotropic mixtures using advanced distillation techniques such as pressure swing distillation, extractive distillation, and hybrid...
-
respond to the discussion In the IMA article, Activity-Based Costing (ABC) is explained as a cost allocation technique that traces indirect costs (often referred to as "overhead") to specific cost...

Study smarter with the SolutionInn App


