Calculate the average CH bond enthalpy in methane using the data tables. Calculate the percent error in
Question:
Table 4.3
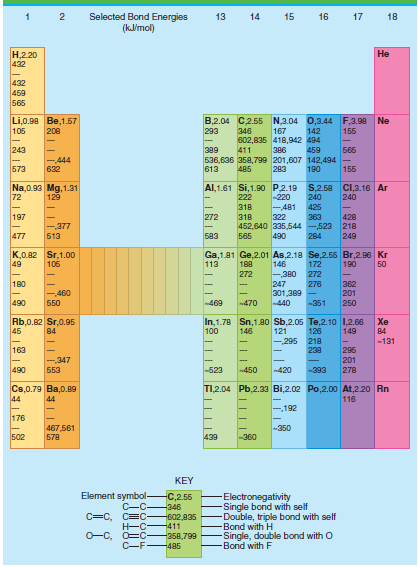
Transcribed Image Text:
Selected Bond Energies (klmol) 13 14 15 16 17 18 Н.220 432 Не 432 459 565 B,2.04 C2.55 N,3.04 0,3.44 F,3.98 Ne Li,0.90 Be,1.57 105 208 293 346 167 142 155 602,835 418,942 494 243 Б65 389 411 386 459 -444 536,636 358,799 201,607 142,494 155 573 632 613 485 283 190 Al,1.61 Si,1.90 P.2.19 S,2.58 C,3.16 Ar Na,0.93 Mg, 1.31 72 129 222 -220 240 240 -481 322 363 452,640 335,544 -523 218 318 425 197 272 318 428 -377 477 583 249 513 565 490 284 K,0.82 Sr,1.00 49 Ga,1.81 Ge,2.01 As,2.18 Se,2.55 Br,2.96 Kr 146 106 113 188 172 190 50 272 -380 272 180 247 362 201 250 276 301,389 - -440 -460 490 Б50 -469 470 -351 In,1.78 Sn,1.80 Sb,2.05 Te,2.10 1,2.66 121 -295 Rb,0.82 Sr,0.95 45 Xe 100 149 84 146 126 84 218 238 -131 163 296 201 278 -,347 553 490 -523 -420 450 -393 TI,2.04 Pb,2.33 Bi,2.02 Po,2.00o At,220 Rn Cs,0.79 Ba,0.89 44 116 44 192 176 -350 467,561 578 502 439 -360 KEY Element symbol- C-C C=C, C=C Electronegativity Single bond with self Double, triple bond with self Bond with H Single, double bond with O -Bond with F C,2.55 -346 602,835 -411 O-C, O=C- -358,799 C-F485
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 42% (7 reviews)
CH 4 gCg 4Hg H o R 4H o f H g H o f C g H o f CH ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Average bond enthalpies are generally defined for gas-phase molecules. Many substances are liquids in their standard state. By using appropriate thermo-chemical data from Appendix C, calculate...
-
Given the data in Table 4.1 (Appendix B, Data Tables) and the following information, calculate the single bond enthalpies and energies for SiF, SiCl, CF, NF, OF, HF: HF(g) SiF,(g) SiCl,(g) CF,(g)...
-
Nitrogen is a vital component of proteins and nucleic acids, and thus is necessary for life. The atmosphere is composed of roughly 80% N2, but most organisms cannot directly utilize N 2 for...
-
Jim Redman purchased a car from Bill Branch Chevrolet. However, the car had been reported stolen and Jim was unable to secure a good title. Redman sued Bill Branch Chevrolet on the breach of the...
-
Vision statements should be massively inspiring, overarching, and long term. a) Provide several examples of potential vision statements for various organizations and discuss how such vision...
-
The general ledger trial balance of Jojoba Ltd includes the following asset accounts at 30 June 2023. Jojoba Ltds investments are part of a long-term investment strategy. The company classifies...
-
The car bumper is made of polycarbonate polybutylene terephthalate. If \(E=2.0 \mathrm{GPa}\), determine the maximum deflection and maximum stress in the bumper if it strikes the rigid post when the...
-
On January 1, 2012, Richards Inc. had cash and common stock of $60,000. At that date, the company had no other asset, liability, or equity balances. On January 2, 2012, it purchased for cash $20,000...
-
Dymac Appliances uses the periodic inventory system. Details regarding the inventory of appliances at January 1, purchases invoices during the next 12 months, and the inventory count at December 31...
-
Montreal Electronics Company manufactures two large-screen television models: the Nova, which has been produced for 10 years and sells for $900, and the Royal, a new model introduced in early 20x0,...
-
Show what reagents you would use to prepare each of the following ethers via a Williamson ether synthesis, and explain your reasoning. a. b. c. OMe
-
The heat capacity of α -quartz is given by The coefficient of thermal expansion is given by β = 0.3530 à 10 4 K 1 and V m = 22.6 cm 3 mol+. Calculate ÎS m for...
-
Copy and complete the statement with sometimes, always, or never. Sketch examples to justify your answer. If two lines are not coplanar, then they? intersect.
-
Identify the two types of class social scientists study and articulate how they intersect .
-
1. Rewrite each equation in logarithmic form. a) 43 = 64 b) 128 = 27 c) 5-2 d) () = 1 25 = 0.25 e) 6* = y f) 10 = 100 000 g) 27 h) v=b" 3-3
-
Discus how access and equity principles relate to people with disadvantages in the community sector .
-
Gillian is the manager of a nursing home with a diverse workforce, in regards to qualification, age, experience and enthusiasm. Her most senior staff, Mary, is very competent, enthusiastic and worked...
-
1. A 220 ml concentrated hydrochloric acid solution is 26,42% HCI and 73.58% water by mass. The specific gravity of the solution is 1.165. a. What mass of pure HCI is contained in this solution? b....
-
Identify and then graph each conic section. If it is a parabola, give the vertex, focus, directrix, and axis of symmetry. If it is an ellipse, give the center, vertices, and foci. y + 4 = (x + 3) 2
-
What is your opinion of advertising awards, such as the Cannes Lions, that are based solely on creativity? If you were a marketer looking for an agency, would you take these creative awards into...
-
What is the resonance frequency of a proton in a magnetic field of 14.1 T?
-
33S has a nuclear spin of% and a nuclear g-factor of 0.4289 calculate the energies of the nuclear spin states in a magnetic field of7.500 T.
-
Calculate the frequency separation of the nuclear spin levels of a 14N nucleus in a magnetic field of 15.4 T given that the magnetogyricrati ratio is 1.93 X 107 T-1 s-l
-
Can you draw a graph to show me how I would depict a change in the Monocentric City Model due to an inflow on the shape of Northwern cities due to the mass migration of African-Americans from the...
-
The firm's production function tell you if a twofold (threefold, etc) increase in all inputs increases output by less than twofold (less than threefold, etc.). Based on this information answer the...
-
After participating in the Application Based Activity (ABA) on the case involving allegations of fraud against the owner of a dating website, I judged the case in favor of the defendant. My decision...

Study smarter with the SolutionInn App


