The following compound is one of the strongest known acids: (a) Explain why it is such a
Question:
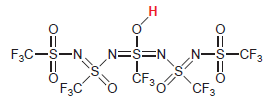
(a) Explain why it is such a strong acid.
(b) Suggest a modification to the structure that would render the compound even more acidic.
Transcribed Image Text:
Н II N-S-CF3 N=S=N F3C-S-N. `CF3 O: F3C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
a When the proton is removed the resulting conjugate base is highly resonance stabil...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following compound is optically inactive. Explain why.
-
Which nitrogen atom in the following compound is more basic?Explain.
-
The following compound is an intermediate in the biosynthesis of one of the twenty common a-amino acids. Which one is it likely to be, and what kind of chemical change must take place to complete...
-
Let be twice-differentiable and one-to-one on an open interval I. Show that its inverse function g satisfies When is increasing and concave downward, what is the concavity of -1 = g? g"(x) =...
-
Longview Baskets has in its inventory 2,000 damaged baskets that cost $20,000. The baskets can be sold in their present condition for a total of $12,000, or repaired at a cost of $7,000 and sold for...
-
The brakes do a certain amount of work to stop a car that is moving at a particular speed. How much work must the brakes do to stop a car that is moving four times as fast?
-
You should have found that the confidence interval when utilizing the paired structure of the data was narrower than the confidence interval ignoring this structure (this will generally be the case,...
-
Tanaka Manufacturing Company is considering three new projects, each requiring an equipment investment of $22,000. Each project will last for 3 years and produce the following cash inflows. The...
-
In a 150-250 word response (excluding references) and respond to any two of the following bullet points in your response. Describe the different stages a proposed product will need to go through...
-
A manufacturer has two different factories that produce three grades of steel: structural steel, rail steel, and pipe steel. They must produce 32 tons of structural, 26 tons of rail, and 30 tons of...
-
For each pair of compounds below, predict which will be more acidic: (a) HCl HBr (b) H 2 O H 2 S (c) NH 3 CH 4 (d) (e) Cl,C Cl3
-
Amphotericin B is a powerful antifungal agent used for intravenous treatment of severe fungal infections. Identify the most acidic proton in this compound: , Amphotericin B NH2
-
Identify the primary market functions of investment bankers.
-
The hurricane sandy case study mentions two policies: state government laws of controlling prices artificially so all have access to gas and rationing through license plates restrictions on alternate...
-
How do you theorize the impact of government intervention and its influence on free market principles? Specifically, how does this balance affect national competitiveness and the attraction of...
-
Aniyah earned $25,000 as an early childhood teacher in the Lindbergh School District. She was promoted to a sixth grade teaching position that included an 80% raise. What will she earn as a new sixth...
-
You have been working on a group project with several other students. The project has generally been going well, but one person on the project has not been pulling his or her weight, relying on...
-
A firm's profit increased from 2010 to 2011 by 20%, but it decreased by 17% from 2011 to 2012. (a) (5 points) Which of the years 2010 and 2012 had the highest profit? (b) (5 points) What percentage...
-
Use the evenodd properties to find the exact value of each expression. Do not use a calculator. cos(-270)
-
At the beginning of its fiscal year, Lakeside Inc. leased office space to LTT Corporation under a seven-year operating lease agreement. The contract calls for quarterly rent payments of $25,000 each....
-
Write structures for the products that would be formed under the conditions in Problem 11.34 if cyclopentanol had been used as the starting material. If more than one product can reasonably be...
-
Starting with isobutane, show how each of the following could be synthesized. (You need not repeat the synthesis of a compound prepared in an earlier part of this problem.) (a) tert-Butyl bromide (b)...
-
Outlined below is a synthesis of the gypsy moth sex attractant disparlure (a pheromone). Give the structure of disparlure and intermediates A-D. NaNH2 1-bromo-5-methylhevane A (CHi) liq NHa HC CNa...
-
Compare life cover end funeral cover stating the advantages and disadvantages of each.
-
What are the change management issues in C 3 solutions?
-
What are 2 key events discussed in the documentary kanyi?

Study smarter with the SolutionInn App


