Amphotericin B is a powerful antifungal agent used for intravenous treatment of severe fungal infections. Identify the
Question:
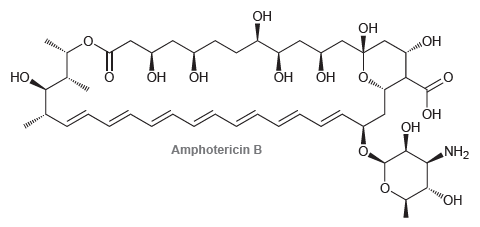
Transcribed Image Text:
ОН ОН Он он ОН ОН ОН Но, ОН Он н Amphotericin B NH2 ОН
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (15 reviews)
HO OH ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Identify the most acidic site in thesecompounds: NH2 CH.COCH,CH,CH3 c) a) b) CH3 e) CH;CH,CH,COH d) CH,CH,CCH,CH3
-
Identify the most acidic proton in each of the following compounds: Xo Xx
-
Identify the most acidic hydrogen in each of these compounds: a) HOCCH,CH,SOH 0 CO H 0 I e) CHCCHCOCHCH3 0 b) CH-CH,CH,C=N d) 0 CHOH f) HNCHCOH
-
Show that is one-to-one and find f(x) = J2 1 + 1 dt
-
Locate the annual reports (10-K) of McDonalds Corporation (MCD) for the most recent three years reported (2013, 2014, &2015). For this assignment, do NOT use the Dupont Method (if applicable). Using...
-
Write a short note on Global Warming.
-
Britfly describe the meaning of the terms (a) safeguarding of assets, (b) reliability of the financial records, and (c) transactions as these terms relate to internal accounting control.
-
Amber Company produces electronic fish finders. It makes two different fish finders: the standard model, which is produced in bulk and sells for $200, and the deluxe model, which comes in various...
-
Does an excellent Employee Value Proposition make their employees want to stay with a company for a long time? how
-
The Future Government Club wants to sponsor a panel discussion on the upcoming national election. The club wants four of its members to lead the panel discussion. Obtain a simple random sample of...
-
The following compound is one of the strongest known acids: (a) Explain why it is such a strong acid. (b) Suggest a modification to the structure that would render the compound even more acidic. II...
-
Predict the position of equilibrium for each of the following reactions: (a) (b) (c) HO NH NH2
-
If a project begins to fall behind, what can a project manager do to get the project back on schedule?
-
a) How can the corporate finance discipline help a Chief Financial Officer (CFO) of acompany in performing his or her job responsibilities? (4 marks) b)Discuss the three main areas of concern of...
-
Who should be allowed to access a financial service provider's customer file?
-
Your client is the U.S. shoe producing company. It wants to establish a manufacturing center in Somalia. Discuss the client's proposal (consider the costs, political risks, geographical location,...
-
Assume the M&M assumptions with taxes hold. The tax rate is equal to 20%, the risk-free rate is equal to 1%, and the market risk premium is equal to 6%. Firms A and B are identical except for their...
-
A firm has an initial debt-to-capital ratio of 10% and is planning to get rid of all of its debt. The tax rate is 30% and the firm's initial pre-tax cost of debt is equal to 5%. What is the firm's...
-
What is the matrix equation form of the following system? 6x + 3y = 9 5x - y = 4 Provide a proof for each of the following.
-
Catalytic hydrogenation of naphthalene over PdC results in rapid addition of 2 moles of H 2 . Propose a structure for this product.
-
Provide the reagents necessary for the following syntheses. More than one step may be required. (a) (b) (c) (d) (e) (f) HO HO Br OH HO HO CI OH
-
Predict the major product from each of the following reactions. (a) (b) (c) (d) (e) (f) OH SOC, pyr OH HBr NaNH2 OH OH (1) TsCI, pyr (2) EtSNa Nal, H2SO OH
-
The following reaction does not produce the product shown. (a) Predict the major product from the conditions shown above, and write a detailed mechanism for its formation. (b) What reaction...
-
Whole blood has a surface tension of 0.058 N/m and a density of 1,050 kg/m. To what height can whole blood rise in a capillary blood vessel that has a radius of 2.3 x 10-6 m if the contact angle is...
-
A large helium filled balloon is used as the center piece for an advertising display. The balloon alone has a mass of 225 kg and it is filled with helium gas until its volume is 326 m.
-
what ways does organizational culture, encompassing values, norms, and leadership styles, influence team cohesion, morale, and productivity, and how can organizations cultivate a culture that...

Study smarter with the SolutionInn App


