Treatment of catechol with formaldehyde in the presence of an acid catalyst produces a compound with molecular
Question:
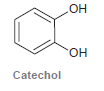
Transcribed Image Text:
HO он Catechol
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (14 reviews)
OH...View the full answer

Answered By

Mary Boke
As an online tutor with over seven years of experience and a PhD in Education, I have had the opportunity to work with a wide range of students from diverse backgrounds. My experience in education has allowed me to develop a deep understanding of how students learn and the various approaches that can be used to facilitate their learning. I believe in creating a positive and inclusive learning environment that encourages students to ask questions and engage with the material. I work closely with my students to understand their individual learning styles, strengths, and challenges to tailor my approach accordingly. I also place a strong emphasis on building strong relationships with my students, which fosters trust and creates a supportive learning environment. Overall, my goal as an online tutor is to help students achieve their academic goals and develop a lifelong love of learning. I believe that education is a transformative experience that has the power to change lives, and I am committed to helping my students realize their full potential.
5.00+
4+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Hexachlorophene, a substance used in the manufacture of germicidal soaps, is prepared by reaction of 2, 4, 5-trichiorophenol with formaldehyde in the presence of concentrated sulfuric acid. Propose a...
-
Delrin (polyoxymethylene) is a tough self-lubricating polymer used in gear wheels. It is made by polymerizing formaldehyde in the presence of an acid catalyst. a. Propose a mechanism for formation of...
-
The industrial synthesis of methyl tert-butyl ether involves treatment of 2-methylpropene with methanol (CH3OH) in the presence of an acid catalyst, as shown in the following equation. CH3 H3C H3C...
-
A medical research study on a new medicine for multiple sclerosis is being conducted with 24 patients. After the study was concluded, it was determined that 16 patients reacted favorably to the...
-
Please discuss any special accounting treatments necessary to be aware of when disposing of part or all of an investment in a subsidiary, particularly address whether there are any affects of...
-
The \(x\) component of the final velocity of the standard cart in Figure 4.8 is positive. Can you make it negative by adjusting this cart's initial speed while still keeping the half cart initially...
-
Determine the slope at \(A\). The A992 steel beam has a moment of inertia of \(I=125\left(10^{6} ight) \mathrm{mm}^{4}\). 18 kNm A -4 m- D B3m 3m 4m m- 18 kNm
-
The Great North Woods Clothing Company is a mail-order company that processes thousands of mail and telephone orders each week. They have a customer service number to handle customer order problems,...
-
l 5. Jane and Herbert were in a relationship but have since broken up. Jane decided that she would enter Herbert's house to take what she believed was hers, an engagement ring, which Jane had given...
-
In August 2010, Michael Dell, Dell Inc.'s CEO and chairman of the board, was reelected to Dell's board of directors by Dell's shareholders. However, not all of the shareholders were happy with Mr....
-
Devise an efficient synthesis for the following transformation (recall that aldehydes are more reactive than ketones): H.
-
Predict the major product(s) for each reaction below. (a) (b) (c) 1) LAH 2) , 1) PhMgBr 2) H,0
-
Cabrera Company produces a variety of chemicals. One division makes reagents for laboratories. The division's projected income statement for the coming year is: Sales (384,000 units @...
-
When it comes to costume design, why is it important to focus on the silhouette of a costume? Explain.
-
Checker Restaurant Inc. has two divisions, Canadian and European. The European division had assets of $32,000,000. Checker has a cost of capital of 9.0% overall. During the most recent year, revenues...
-
On September 1, Global Tech Inc. purchased merchandise for resale for $20,800 on credit terms 2/15, n/60 using the gross method and a perpetual inventory system. Global Tech incurred a shipping...
-
Assume that your advertising agency is compensated based on markup percentage charges. If the cost of the advertising is $850,000, and the agency includes 15% markup, how much will the client be...
-
Calculate the accounting impact on Google if it implemented a new stock grant plan with the following characteristics: Grant of 1000 shares. The shares vest in equal proportions over a 3-year period...
-
Solve each system using the inverse of the coefficient matrix. 5x - 4y = 1 x + 4y = 3
-
Coastal Refining Company operates a refinery with a distillation capacity of 12,000 barrels per day. As a new member of Coastal's management team, you have been given the task of developing a...
-
Designate the (R) or (S) configuration at each chirality center in the following molecules. Cl SH Br Br
-
Albuterol, shown here, is a commonly prescribed asthma medication. For either enantiomer of albuterol, draw a three-dimensional formula using dashes and wedges for bonds that are not in the plane of...
-
(a) Write the structure of 2 ,2-dichlorobicyclo [2.2.1] heptane. (b) How many chirality centers does it contain? (c) How many stereoisomers are predicted by the 2n rule? (d) Only one pair of...
-
Given the data below answer the question. E F G H J Inventory Turnover Ratio Ending Inventory (Dec. 31st) $50,000 Month Ending Inventory Cost of Goods Sold Inventory Turnover Ratio Formulas January...
-
[3] 4. In 1995, Mary bought an antique chair for $55. It is estimated that the chair will increase in value by 1.8%/year. a) Find the current value of the chair. 121 [2] b) In what year will the...
-
Outback Outfitters sells recreational equipment. One of the company s products, a small camp stove, sells for $ 1 4 0 per unit. Variable expenses are $ 9 8 per stove, and fixed expenses associated...

Study smarter with the SolutionInn App


