Using acetonitrile (CH 3 CN) and CO 2 as your only sources of carbons, identify how you
Question:
a.
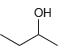
b.
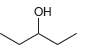
c.
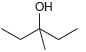
d.
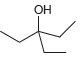
Transcribed Image Text:
Он ОН
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
a b c d CH3CN H3O ...View the full answer

Answered By

Krishnavendra Y
I am a self motivated financial professional knowledgeable in; preparation of financial reports, reconciling and managing accounts, maintaining cash flows, budgets, among other financial reports. I possess strong analytical skills with high attention to detail and accuracy. I am able to act quickly and effectively when dealing with challenging situations. I have the ability to form positive relationships with colleagues and I believe that team work is great key to performance. I always deliver quality, detailed, original (0% plagiarism), well-researched and critically analyzed papers.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Name the following compounds: a. b. c. d. e. f. CH3 CH3 CH2CH3 CH3 HN CH3 CH3 CH3 CH-CH3
-
Name the following compounds: a. b. c. d. e. f. g. h. i. COOH Br Br Br Br CH3 ,. CHCH NO. OCH, CH2CH3 SOH CI CI CH3 CH2CH3 Cl
-
Give the systematic name for each of the following compounds: a. b. c. d. e. CH CH,CHCH,CH CHCH NHCH3 CH3 CH3 CH CH-CHCH2CHCH CH3 HCH3 H2CH3 CH CHCHCH,CH CH CI CH CH2CH2CH2CHCH2CH2CH2CHa CH,CCH2CH3...
-
In Exercises 7681, find the domain of each function. g(x) = 4 x - 7
-
In discussing future actions regarding strengths, the goal is to come up with strategies that help us build on and benefit from our strengths more than before. In discussing your future actions, you...
-
Is intellectual property theft an ethical issue? Evaluate the concept of duty and its implications for the integrity of intellectual property rights.
-
Apex Electrical Supply, Inc., purchased inventory for \($2,000\) and also paid \($125\) freight to have the inventory delivered. Apex Electrical Supply, Inc., returned \($500\) of the goods to the...
-
The following merchandise transactions occurred in December. Both companies use a perpetual inventory system. Dec. 3 Pippen Company sold merchandise to Thomas Co. for $32,000, terms 2/10, n/30, FOB...
-
You are planning for an early retirement, so you decide to invest $5,000 per year, starting at age 23. You plan to retire when you accumulate $1,000,000. If the average rate of return on your...
-
If the consumption function is given by C = 4200 + 0.75Y, state the marginal propensity to consume and deduce the marginal propensity to save.
-
Identify the number of Ï electrons in each of the following compounds. a. b. c. d. e. N'
-
Compound A has molecular formula C 9 H 8 O 2 and exhibits a strong signal at 1740 cm -1 in its IR spectrum. Treatment with two equivalents of LAH followed by water gives the following diol. Identify...
-
1. What micro environmental factors affected both the first generation and second generation models of the Toyota Prius? How well has Toyota dealt with these factors? 2. Outline the major macro...
-
Consider the statement Customer complaints are customer opportunitiesbut only if we know about them. Do you agree or disagree? Why?
-
Comment on the following field situations and make recommendations for corrective action. a. One of the interviewers has an excessive rate of refusals in in-home personal interviewing. b. In a CATI...
-
A deep pool of water is initially at \(4^{\circ} \mathrm{C}\) and suddenly the surface is exposed to a freezing front at \(-4{ }^{\circ} \mathrm{C}\) and remains so exposed for a long time. Find the...
-
What are the key factors behind a successful report presentation?
-
You have been asked by an investor to value a restaurant. Last year, the restaurant earned pretax operating income of \($300,000.\) Income has grown 4% annually during the past 5 years, and it is...
-
How are auditors judgments about the quality or effectiveness of internal control affected by anchoring?
-
Consider a closed, rigid tank with a volume of 0.8L, filled with cold water initially at 27C. The tank is filled such that there are no voids (air pockets) within. The initial pressure within the...
-
Explain how you would expect the diagram of potential energy versus dihedral angle about the C2-C3 (central) carbon-carbon bond of 2,2,3,3-tetramethylbutane to differ from that for ethane (Fig. 2.3),...
-
When the structure of compound A was determined in 1912, it was found to have an unusually long C-C bond and unusually large C-C-C bond angles, compared with the similar parameters for compound B...
-
When the structure of compound A was determined in 1912, it was found to have an unusually long C-C bond and unusually large C-C-C bond angles, compared with the similar parameters for compound B...
-
9- Assume that printer interrupt priority: 2, disk interrupt priority:4, serial line interrupt priority: 6, Ethernet interrupt priority: 5. Determine the order of interrupt services according to: A...
-
4- Assume that printer interrupt priority: 2, disk interrupt priority:4, Ethernet interrupt priority: 5. A user program starts at t = 0 and takes 40 seconds to complete. At t = 10, a disk interrupt...
-
Robbie is a 3 year old boy in the bur oak room at little.ly early learning centre. You have been one of robbies educators for the past two weeks. You and your colleagues have been concerned about...

Study smarter with the SolutionInn App


