Using acetylene and methyl bromide as your only sources of carbon atoms, propose a synthesis for each
Question:
(a)
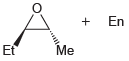
(b)
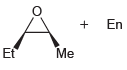
Transcribed Image Text:
En Et Me En Et Me
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
a b 1 H Lindlars ca...View the full answer

Answered By

Felix Mucee
I am a detailed and thorough professional writer with 5 years of administrative experience- the last 2 years in academic writing and virtual office environment. I specialize in delivering quality services with respect to strict deadlines and high expectations. I am equipped with a dedicated home office complete with a computer, copier/scanner/fax and color printer.
I provide creative and detailed administrative, web search, academic writing, data entry, Personal assistant, Content writing, Translation, Academic writing, editing and proofreading services. I excel at working under tight deadlines with strict expectations. I possess the self-discipline and time management skills necessary to have served as an academic writer for the past five years. I can bring value to your business and help solve your administrative assistant issues.
4.70+
13+ Reviews
33+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Dimethoxyethane (DME) is a polar aprotic solvent often used for S N 2 reactions. Propose a plausible synthesis for DME using acetylene and methyl iodide as your only sources of carbon atoms....
-
Using acetylene and 2-methylpropane as your only sources of carbon atoms, propose a plausible synthesis for 4-methyl-2-pentanone. You will need to utilize many reactions from previous chapters.
-
Using acetylene and ethylene oxide as your only sources of carbon atoms, propose a synthesis for each of the following compounds. a. b.
-
Return to Better Mousetraps in Problem 18. Suppose the firm can cut its requirements for working capital in half by using better inventory control systems. By how much will this increase project NPV?...
-
What would be the advantage to an exporter of buying a futures contract on the foreign exchange rate value of the dollar?
-
Suppose a player has a batting average over many games of 0.200 (hes not very good). In his next game, he goes 2 for 4, which is a batting average of 0.500 for the game. Does it follow that his new...
-
For an ideal solution, the enthalpy change of mixing \(\left(\Delta H_{\text {mix }} ight)\) is always given by (a) \(\Delta H_{\text {mix }}=1\) (c) \(\Delta H_{\text {mix }}=-1\) (b) \(\Delta...
-
The comparative balance sheets of Incloud Airlines show the following information for a recent year (amounts in thousands of US$): aCash was $378,511 at the beginning of the year and $418,819 at the...
-
How can organizations develop and implement effective diversity and inclusion strategies that not only improve representation but also enhance overall organizational performance and creativity?
-
The following table provides the information necessary to construct a project network and project crash data: a. Construct the project network. b. Manually crash the network to 28 weeks. c. Formulate...
-
A bond that pays interest forever and has no maturity date is a perpetual bond. How is the yield to maturity on such a bond determined?
-
Draw a mechanism and predict the major product for each reaction. a. b. c. d. e. f. ? 1) LAH H. 2) H20
-
From the two graphs below, construct two graphs that depict the cost behaviour on a perdriver-unit basis. Which of the two constructed graphs shows fixed cost behaviour? Variable cost behaviour?...
-
As Health care administrators and advocates of patients how do we ensure that care is not substandard based on assumptions? Why do you think, no one intervened and advocated for Henrietta Lacks? ...
-
Does free trade benefit all countries involved, or create winners and losers? Explain with at least one example. What is protectionism? Describe some protectionist measures described in the video....
-
1) Find a good definition of Public Relations 2) What is the name of a national society you could join if you were to be a Public Relations professional 3) Find an example of a company that wrote an...
-
Hobbes, the stuffed tiger, has a mass of 31.8 kg. Calvin, the little boy, has a mass of 25.1 kg. In a game of football, Hobbes runs at Calvin at 11 m/s. Calvin is running in the same direction as...
-
How does the net debt as a percentage of total capitalization ratio differ from the debt-to-equity ratio?
-
Factor the trinomial, if possible. 6k 2 + 5kp - 6p 2
-
1. Firms may hold financial assets to earn returns. How the firm would classify financial assets? What treatment will such financial assets get in the financial statements in accordance with US GAAP...
-
Identify the reagents a?f in the following scheme: .Br CH-
-
Galactose, a constituent of the disaccharide lactose found in dairy products, is metabolized by a pathway that includes the isomerization of UDP-galactose to UDP-glucose, where UDP = uridylyl...
-
Propose a structure consistent with the following spectral data for a compound C8H18O2: IR: 3350 cm1. 1H NMR: 1.24 (12 H, singlet); 1.56 (4 H, singlet); 1.95 (2 H, singlet)
-
Go into depth about the topic: Do women in the workplace still have to tolerate sexual harassment to advance or maintain their job positions? Why or why not? THE SCENARIO: You are human resource...
-
A pile group consists of nine friction piles in clay soil (see Figure 10-40). The diameter of each pile is 16 in., and the embedded length is 30 ft each. Center-to-center pile spacing is 4 ft. Soil...
-
Honest to Goodness Merchandising was registered as a Single Proprietorship by May Mendoza, the owner in the Department of Trade and Industry. Selected transactions for the month of July, 2019 are...

Study smarter with the SolutionInn App


