Using formaldehyde and acetaldehyde as your only sources of carbon atoms, show how you could make each
Question:
(a)
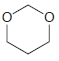
(b)
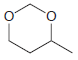
(c)
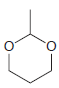
(d)
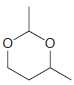
Transcribed Image Text:
O.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (18 reviews)
a b c d 1 ...View the full answer

Answered By

Keziah Thiga
I am a self motivated financial professional knowledgeable in; preparation of financial reports, reconciling and managing accounts, maintaining cash flows, budgets, among other financial reports. I possess strong analytical skills with high attention to detail and accuracy. I am able to act quickly and effectively when dealing with challenging situations. I have the ability to form positive relationships with colleagues and I believe that team work is great key to performance. I always deliver quality, detailed, original (0% plagirism), well-researched and critically analyzed papers.
4.90+
1504+ Reviews
2898+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Using toluene and acetylene as your only sources of carbon atoms, show how you would prepare the following compound.
-
Using acetylene and ethylene oxide as your only sources of carbon atoms, propose a synthesis for each of the following compounds. a. b.
-
Using acetylene and methyl bromide as your only sources of carbon atoms, propose a synthesis for each of the following compounds: (a) (b) En Et Me En Et Me
-
The accompanying data are consistent with summary statistics that appeared in the paper Shape of Glass and Amount of Alcohol Poured: Comparative Study of Effect of Practice and Concentration (...
-
Domremy Drywall Inc. needs to purchase new equipment to better service its client accounts. The cost of the equipment is $175,000. It is estimated that the firm will save $35,000 annually (after tax)...
-
Josef manages a kebab shop in Sydneys central business district and has the following information on the shops demand and costs: a. To maximize profit, how many kebabs should Josef sell per hour,...
-
Information from Sheppard Picture Frames cash account as well as the November bank statement is presented next. Requirements 1. Prepare the bank reconciliation on November 30. 2. Prepare all...
-
CVP analysis (CMA, adapted) Galaxy Disks projected operating income for 2008 is $200,000, based on a sales volume of 200,000 units. Galaxy sells disks for $16 each. Variable costs consist of the $10...
-
What experiences have you had working in an environment where wearing protection was required? Did you wear personal protective equipment (PPE) willingly? Could the type of PPE provided have been...
-
A Global private bank is aggressively looking to leverage technology to improve customer experience and reduce operational costs. Over the last few years, it has tied up with at least five startups...
-
Identify the reagents necessary to produce each of the following compounds via an aldol reaction. (a) (b) (c) (d) (e)
-
Draw a mechanism for the following transformation: NaOH, heat
-
The Washington, D.C. police suspected Antoine Jones of being a drug dealer. Without a valid search warrant, they attached a GPS tracking device to his car. For 28 days, they used the GPS to determine...
-
What are 3 approaches for representing anOPERATING ENVIRONMENT to a model or simulation?
-
What is a mock-up?
-
What sampling methods are available for substantive testing, and under what conditions should each be used?
-
How do SEs employ mock-ups? Provide examples and explain what types of knowledge SEs would expect to gain from them?
-
M&S has a number of challenges and issues SEsmust be prepared to address. Identify the 7 challenges addressed in the chapter.
-
A stock has had the following year-end prices and dividends: What are the arithmetic and geometric returns for the stock? Price Year Dividend $64.18 $0.57 71.05 76.85 0.62 4 63.12 0.68 72.81 0.77...
-
A red card is illuminated by red light. What color will the card appear? What if its illuminated by blue light?
-
Compound A, C6H12O2, was found to be optically active, and it was slowly oxidized to an optically active carboxylic acid B, C6H12O3, by +Ag(NH3)2. Oxidation of A by anhydrous CrO3 gave an optically...
-
Remembering that a protonated aldehyde or ketone is a type of carbocation, and that carbocations are electrophiles, propose a curved-arrow mechanism for the electrophilic aromatic substitution...
-
From your knowledge of the reactivity of LiAlH4, as well as the reactivity of epoxides with nucleophiles, predict the product (including stereochemistry, if appropriate) in each of the following...
-
A 0.8 liter of boiled water at 100C is poured into an adiabatic mug in an air-conditioned hotel room of 25C. An educational Stirling engine is placed onto the mug, whereby it receives the heat from...
-
During the month of February, Riverbed Corp\'s employees earned wages of $ 8 6 0 0 0 . Withholdings related to these wages were $ 6 5 7 9 for FICA, $ 1 5 6 2 0 for federal income tax, and $ 4 1 8 0...
-
Power Drive Corporation designs and produces a line of golf equipment and golf apparel. Power Drive has 1 0 0 , 0 0 0 shares of common stock outstanding as of the beginning of 2 0 2 4 . Power Drive...

Study smarter with the SolutionInn App


