When the following compound is treated with Br 2 in the presence of a Lewis acid, one
Question:
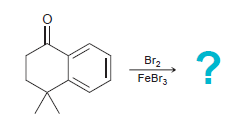
Transcribed Image Text:
Br FeBr3 ?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (8 reviews)
Br...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
What product is formed when the following compound is treated with Ag2O? HO
-
When the following compound is treated with sodium methoxide in methanol, two elimination products are possible. Explain why the deuterated product predominates by about a 7:1 ratio (refer to Problem...
-
Only a substitution product is obtained when the following compound is treated with sodium methoxide: Explain why an elimination product is not obtained. CH3 Br CH3
-
Compare the hoop stress (r,/2) predictions from elasticity with voids given by relation (15.5.18) with the corresponding results from classical theory. Choosing N = 1/2 and L = 2, for the elastic...
-
How can market research benefit a small business owner? List some possible sources of market information.
-
The following information is taken from the trial balance of Petras Pedicure Parlour. Required (a) Given that the cost of the inventory on 30 June 2026 is \($25\) 300, prepare the closing entries on...
-
Investigate the file search facility on your computer. Which type of search method do you think it uses? Why do you think this partic ular search method was chosen? What problems could this approach...
-
An evaporationcrystallization process of the type described in Example 4.5.2 is used to obtain solid potassium sulfate from an aqueous solution of this salt. The fresh feed to the process contains...
-
If we are an auditor, then carry out an audit of the financial statements by December 31, 2022. When we conducted the audit, we received information that the client in November 2022 apparently...
-
Following is the unadjusted trial balance for Alonzo Institute as of December 31, 2017. The Institute provides one-on-one training to individuals who pay tuition directly to the business and offers...
-
Ca(HCO 3 ) 2 (s) decomposes at elevated temperatures according to the stoichiometric equation Ca(HCO 3 ) 2 (s) ???? CaCO 3 (s) + H 2 O(g) + CO 2 (g). a. If pure Ca(HCO 3 ) 2 (s) is put into a sealed...
-
Calculate A for the isothermal compression of 2.95 mol of an ideal gas at 325 K from an initial volume of 60.0 L to a final volume of 20.5 L. Does it matter whether the path is reversible or...
-
The jet water ski has reached its maximum velocity of 70 km/h when operating in salt water. The water intake is in the horizontal tunnel in the bottom of the hull, so the water enters the intake at...
-
Calculate the return on shareholders' equity for Ivanhoe in 2 0 2 4 . Note that Ivanhoe's articles of incorporation authorize only common shares. The average return for the shares listed on the...
-
What statements are intended to meet the needs users who are not in a position to require an entity to prepare reports tailored to their particular information needs?
-
Direct combination costs and stock issuance costs are often incurred in the process of making a controlling investment in another company. How should those costs be accounted for in a pre-2009...
-
The clinic s COO asks you to create a budgeted income statement ( also called a profit and loss statement ) for the clinic for the upcoming fiscal year. She has asked an intern to create a volume...
-
Are there issues associated with keeping the large sum of money in accumulated earnings? The corporation has a large amount of money in retained earnings. The corporations retained earnings...
-
(a) Use the Midpoint Rule for double integrals with m = n = 2 to estimate the area of the surface z = xy + x 2 + y 2 , 0 < x < 2, 0 < y < 2. (b) Use a computer algebra system to approximate the...
-
Use critical values to test the null hypothesis H0: 1 2 = 20 versus the alternative hypothesis H0: 1 2 20 by setting a equal to .10, .05, .01, and .001. How much evidence is there that the...
-
Rationalize each of the following observations by postulating a structure for the fragment ion(s) and the mechanisms for their formation. The EI mass spectrum of 2-methoxybutane shows a base peak at...
-
What is the wavelength of light with a wave-number of 1720 cm-1?
-
Predict the relative intensities of the three peaks in the mass spectrum of dichloromethane at m/z = 84, 86, and 88.
-
If you were to enter the banking industry, you might find yourself approving or not approving loans. The following is a good example of a common event you could encounter. Company X is looking for...
-
You see a two year bond with annual coupon rate of 8% that has these flows. Years ahead Cashflows 0 ?? 0.25 0 0.5 0 0.75 1 1.25 1.5 1.75 2 8 0 0 0 108 You will be asked to calculate its price at...
-
Use synthetic division and the Remainder Theorem to evaluate P(c). P(x) = 6x + 5x +9, c = 11/1 P(1) =

Study smarter with the SolutionInn App


