What product is formed when the following compound is treated with Ag2O? HO
Question:
What product is formed when the following compound is treated with Ag2O?
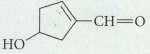
Transcribed Image Text:
HO
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
Silver I oxide oxidizes t...View the full answer

Answered By

Vikas NA
I have 1 year teaching experience in various field such as mathematics, computer science, chemistry. Mathematics is my most interesting subject. I have work as a private tutor in aakash institute for mathematics subject for 0.5 years. then, i was move to other coaching center. I was work in Surya shiksha mandir coaching center as a chemistry tutor for 2 months. After that, i joined coding blocks institute as a computer science tutor and i teach c++, java, python, matlab, database, operating system. I worked 5 month in this institute. My overall teaching experience is very good, student give me very good reviews.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
When the following compound is treated with sodium ethoxide, two condensation products are obtained, both of which are produced via Dieckmann cyclizations. Draw both products. OEt Eto
-
When the following compound is treated with concentrated HCl at 100ºC for several hours, hydrolysis occurs, producing one of the 20 naturally occurring amino acids. Identify which one. H,N,
-
When the following compound is treated with excess methyl iodide, a quaternary ammonium salt is obtained that bears only one positive charge. Draw the structure of the quaternary ammonium salt. *NH2
-
Ag Bio Tech (ABT) was organized on January 1, 2013, by four friends. Each organizer invested $10,000 in the company and, in turn, was issued 8,000 shares of common stock. To date, they are the only...
-
1. A foundation pledges to donate $1 million to an art institute one year in the future. When, and in what amount, should the institute recognize revenue? The institute applies a discount rate of 10...
-
What do you mean by Non-current Liabilities?
-
Consider the thin-walled tube of Fig. 5-26. Use the formula for shear stress, \(\tau_{\text {avg }}=T / 2 t A_{m}\), Eq. 5-18, and the general equation of shear strain energy, Eq. 14-11, to show that...
-
Presented below is the balance sheet of Sargent Corporation for the current year, 2012. The following information is presented.1. The current assets section includes: cash $150,000, accounts...
-
You will make a web site that consists of 2 different HTML files and 1 CSS file. The files will be named index.html, page2.html, and assign1.css. Please note the case of these files, Index.html is...
-
The assets, labilities, and equities of Dundee Design Studio have the following balances at December 31, 2024. The Retained Earnings was $31,000 at the beginning of the year. At year end, Common...
-
Draw the structures of all aldehydes or ketones that could in principle give the following product after application of either the Wolff-Kishner or Clemmensen reduction. CH,CH(CH)h
-
Give the product expected (if any) when butyraldehyde (butanal) reacts with each of the following reagents. (a) PhMgBr, then dilute H3O+ (b) LiAlH4 in ether, then H3O+ (c) Alkaline KMnO4, then H3O+...
-
What is the definition of an integrated marketing mix? How might this affect the research a firm conducts?
-
Completely simplify each expression.
-
A store sells yogurt in small, medium, and large sizes. A small yogurt costs $2, a medium costs $3, and a large costs $5. In one hour, the shop sold 27 yogurts for $98. There were five more large...
-
What is the total cost for the customer service category of the value chain?
-
The Sheridan Law Office has the following monthly telephone records and costs: Calls Costs 4,100 $3,135 3,000 2,700 4,300 3,370 6,700 4,883 4,400 3,435 3,800 2,935 Identify the fixed and variable...
-
= A 33.0 kg crate is being push across a horizontal floor with a force F 190 N that makes an angle 0 = 30.0 with the horizontal as illustrated in the figure below. Find the magnitude of the...
-
Find all x-values where the function is discontinuous. For each such value, give f (a) and lim xa f (x) or state that it does not exist. (x) = 2x 2 - 5x - 3
-
What is the amount of total interest dollars earned on a $5,000 deposit earning 6% for 20 years?
-
Use the information in Figure 4.2 to predict the positions of the equilibria in the reactions in problem 4.4.
-
Draw diagrams like that in Figure 4.3 for the reactions in problem 4.9.
-
Show a free energy versus reaction progress diagram for the following reaction: HCI+ NH3 CI + NH4
-
If the nominal interest rate is 14%, and inflation is 4%, what is the real interest rate? 22.A bond investment yielded 8%. If inflation was 3%, what real return did the bond offer? 23.The following...
-
Bierce Corporation has two manufacturing departments--Machining and Finishing. The company used the following data at the beginning of the year to calculate predetermined overhead rates: Estimated...
-
Discuss how communication channels have changed over the years due to technology.

Study smarter with the SolutionInn App


