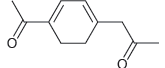
When the following compound is treated with sodium ethoxide, nearly all of it is converted into an
Question:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
This anion is highly stabilized b...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
When the following compound is treated with sodium ethoxide, two condensation products are obtained, both of which are produced via Dieckmann cyclizations. Draw both products. OEt Eto
-
Only a substitution product is obtained when the following compound is treated with sodium methoxide: Explain why an elimination product is not obtained. CH3 Br CH3
-
When the following compound is treated with sodium methoxide in methanol, two elimination products are possible. Explain why the deuterated product predominates by about a 7:1 ratio (refer to Problem...
-
Prove that point B in Figure 18.1 is not Pareto effi cient. Food Fo FB OA Abner E C B Contract curve UAB UBB MB -Medicine Z Belinda Mo Figure 18.1 Edgeworth Box for Exchange; Pareto Efficient Points...
-
Describe the current situation at Bayonne. How might Milliken summarize Bayonne's performance problems?
-
If you were to audit an existing performance-appraisal system, what criteria would you use to judge its effectiveness?
-
Redwing Floor Coverings, Inc., reported the following summarized data at December 31, 2009. Accounts appear in no particular order. Requirement 1. Prepare the trial balance of Redwing Floor Coverings...
-
Martha Jansa executed a will naming her two sons as executors and leaving all her property to them. The will was properly signed and attested to by witnesses. Thereafter, Martha died. When Marthas...
-
a. Project A costs $6,000 and will generate annual after-tax net cash inflows of $2,150 for 5 years. What is the payback period for this Investment under the assumption that the cash Inflows occur...
-
Ricky Corporation had the following alphabetical account balance listing at December 31, 20X1 (in thousands of dollars). Required: Prepare a balance sheet for Ricky Corporation at December 31, 20X1....
-
How have automated work flows tended to impact supply chains?
-
Describe the customer contact points of a favorite restaurant, and discuss whether there should be fewer or more contact points.
-
A company has determined that the cost y to make x (thousand) computer tablets is and that the revenue y from the sale of x (thousand) tablets is Find the break-even point, where cost equals revenue....
-
An electrical engineer has developed a modified circuit board for elevators. Suppose 3 modified circuit boards and 6 elevators are available for a comparative test of the old versus the modified...
-
An engineer wants to compare two busy hydraulic belts by recording the number of finished goods that are successfully transferred by the belts in a day. Describe how to select 3 of the next 6 working...
-
If the probability density of a random variable is given by \[f(x)= \begin{cases}x & \text { for } 0
-
Given the probability density \(f(x)=\frac{k}{1+x^{2}}\) for \(-\infty
-
If the distribution function of a random variable is given by \[F(x)= \begin{cases}1-\frac{4}{x^{2}} & \text { for } x>2 \\ 0 & \text { for } x \leq 2\end{cases}\] find the probabilities that this...
-
What are the elements of negligence that apply in establishing auditor liability under common law?
-
As water moves through the hydrologic cycle, water quality changes are common because of natural phenomena or anthropogenic pollution. Using Figure 11.1, describe how water-quality changes occur...
-
Propose a curved-arrow mechanism for each of the re-actions given in Fig. P17.38. (a) (b) (c) H ,C) ether CH:(CH2)3 CH C CH2 CH3(CH2sC C CH 1-penten-4-yne + Na+:C CH; then allyl bromide- H,C-CH-CH-C...
-
What product(s) would be expected in the same re-action of 3-methyl-4-octyne? Explain.
-
Propose a curved-arrow mechanism for the following reaction. Explain why the equilibrium lies to the right. Ph Ph CH toluenesulfonic acidCH
-
9 1. Compute the indefinite integral. +1 (3+ 4+4) dr.
-
Sarah Jackson Antiques issued its 11%, 10-year bonds payable at a price of $338,220 (face value is $400,000). The company uses the straight-line amortization method for the bond discount or premium....
-
Suppose you start with a full tank of gas (14 gallons) in your truck. After driving 6 hours, you now have 6 gallons left. If x is the number of hours you have been driving, then y is the number of...

Study smarter with the SolutionInn App


