When the following compound is treated with sodium ethoxide, two condensation products are obtained, both of which
Question:
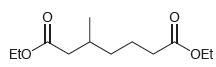
Transcribed Image Text:
OEt Eto
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
When the following compound is treated with sodium ethoxide, nearly all of it is converted into an enolate. Draw the resonance structures of the enolate that is formed, and explain why enolate...
-
What product is formed when the following compound is treated with Ag2O? HO
-
When the following compound is treated with Br 2 in the presence of a Lewis acid, one product predominates. Determine the structure of that product. Br FeBr3 ?
-
find the steady state expression for vo in the circuit fig 9.32 if ig = 500cos2000tmA 3) 9.32 Find the steady-state expression for u,, in the circuit of Fig. P9.32 if i = 500 cos 2000 mA. Figure...
-
Amy is 12 years old now and will attend college at age 18. Her parents plan to fund her college for four years. College costs $20,000 per year as of the time when Amy turns to age 18. If her parents...
-
Nuclear reactors are accompanied by conversion of mass into energy according to the famous equation \(E=m c^{2}\). Hence the mass balance does not hold in such systems. Calculate the mass loss in a...
-
Fill in the Blank. In real-time frequency analysis, the signal is continuously analyzed over all the ___________ bands.
-
A share of a ventures preferred stock is convertible into 1.5 shares of its common stock . The dividend on the preferred stock is $0.50 per share. A. If the firms common stock is currently trading at...
-
2. A journal bearing 4 inches in diameter and 4 inches long has a radial clearance of 0.002 inches. It rotates at 2000 rpm and is lubricated with SAE 10 oil at 200F. In the following, use an applied...
-
A Global private bank is aggressively looking to leverage technology to improve customer experience and reduce operational costs. Over the last few years, it has tied up with at least five startups...
-
Ask students to list three products that seem to have personalities. Describe the personalities. What types of people buy these products? Is there a match between the consumers personality and that...
-
This chapter mentions that psychographic analyses can be used by politicians to market themselves. What are some of the marketing strategies and techniques used by politicians in recent elections?...
-
For each pair of compounds, predict which compound has the higher boiling point. Check Table 6-2 to see if your prediction was right, then explain why that compound has the higher boiling point. (a)...
-
Exercise 9 - 4 A ( Algo ) Inventory turnover LO 0 9 - 0 2 Selected financial information for Solomon Company for Year 4 follows: \ table [ [ Sales , $ 2 , 3 5 0 , 0 0 0 es Exercise 9-4A (Algo)...
-
1 . Today, you can buy a 1 5 - year Treasury bond with a 3 . 5 % coupon rate for $ 1 0 4 7 . 5 5 . Show your work. A ) Calculate the Yield - to - Maturity for this bond B ) Assume the bond is...
-
What major risk to retirement do annuities mitigate? Why is that important?
-
QUESTION 1 - 1 POINT For a Sunday dinner party, each guest can choose from the 7 different kinds of food to eat. If any number of food types can be chosen, how many food type combinations are...
-
Milner Company is working on two job orders. The job cost sheets show the following. Job 201 Job 202 Direct materials $7,500 $9,250 Direct labor 4,000 8,150 Manufacturing overhead 5,050 9,600 Prepare...
-
Use the indicated substitution to convert the given integral to an integral of a rational function. Evaluate the resulting integral. dx 3. ; - 3.
-
All of the following assets can be depreciated, except: (a) A bulldozer (b) A copper mine (c) A surgical robot (d) A conveyor belt
-
How many milliliters of aqueous 0.1 M NaOH are required to form the disodium salt from 100 mg of succinic acid?
-
Give the product(s) formed and the curved-arrow notation for the reaction of 0.01 mole of each reagent below with 0.01 mole of acetic acid. (a) Cs+ -OH (b) H3C--Li (c) NaH
-
Explain why the differences between the first and second pKa values of the dicarboxylic acids become smaller as the lengths of their carbon chains increase (Table 20.3). TABLE 20.3 pK, Values of Some...
-
For each of the following: identify all critical numbers and use these to determine all region(s) where the function is increasing as well as all region(s) where the function is decreasing. Then...
-
Determine the most suitable data structure for the above graph and show it adjacency representation. (5 markah/marks)
-
The table gives the outdoor temperatures observed by a science student on a spring day. Draw a graph of the data, and find the average rate of change of temperature between the following times: a)...

Study smarter with the SolutionInn App


