Would you expect the following compound to be aromatic? Justify your answer. OR N-
Question:
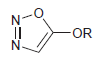
Transcribed Image Text:
OR N-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 81% (16 reviews)
Yes The lone pairs on the nitrogen atoms do not cont...View the full answer

Answered By

Shristi Singh
A freshman year metallurgy and material science student in India.
4.80+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Would you expect the following compound to be aromatic? Explain your answer.
-
How many absorptions would you expect the following compound to have in its 1H and 13C NMRspectra?
-
Do you expect the following compound to be chiral? Explain your answer (consider whether this compound is super-imposable on its mirror image).
-
Solve each system. x + 4y = - 3x + 2y = 17 -9
-
Discuss the role culture plays in organizational change. Include the following: Define organizational culture. Discuss the roots of organizational culture. Discuss the role organizational culture...
-
You decide to open an individual retirement account (IRA) at your local stockbroker that pays 10 percent/year/year for the life of the account. You deposit \($2,000\) today to open the account. For...
-
A quality-control engineer wants to check whether (in accordance with specifications) \(95 \%\) of the electronic components shipped by his company are in good working condition. To this end, he...
-
Suppose you are the manager of a California winery. How would you expect the following events to affect the price you receive for a bottle of wine? a. The price of comparable French wines decreases....
-
The design, layout, and content of a Web site are critical to it being easily found by search engines. It is important to recognize these concepts from your own use and experience of the Web. You...
-
Consider a home theatre system consisting of a television set, a receiver, a DVD player, speakers, and any other components you wish to include. Draw a system diagram for this system. Include both...
-
Do you expect the following dianion to exhibit aromatic stabilization? Explain.
-
Diphenylmethane exhibits two aromatic rings, which achieve coplanarity in the highest energy conformation. Explain. Diphenylmethane
-
Solve Problem 3.33 in Chapter 3 to construct an x-bar chart in conjunction with an R-chart for delivery time at the Great Outdoors Clothing Company. Data in Problem 3.33 in Chapter 3 The Great...
-
Explain briefly the meaning and importance to auditing of the concept of due care.
-
Follett proposes a collaboration between manager and worker whereas Taylor suggests a directive approach. Consider the merits and demerits of each and which will be most appropriate for Industry 4.0...
-
Do any of the five functions and four forms of inventories exist in service firms? If so, which ones, and why? If not, how are the functions served?
-
What capacity is required for a production system that produces 753 good units a year if it has a utilization of 90 percent and yield of 85 percent?
-
The following AON chart was prepared at the beginning of an important software upgrade project. The duration, in days, follows the letter of each activity. What is the critical path? Which activities...
-
What aspect of nuclear fission makes it possible for a chain reaction to occur? What is the difference between a chain reaction in a bomb and one in a nuclear power plant?
-
This problem continues the Draper Consulting, Inc., situation from Problem 12-45 of Chapter 12. In October, Draper has the following transactions related to its common shares: Oct 1 Draper...
-
Give the structure the organic product(s) formed when HBr reacts with each of the following alkenes in the presence of peroxides, and explain your reasoning. If more than one product is formed,...
-
Give the structures of the free radical intermediates in the peroxide-initiated reaction of HBr with each of the following alkenes. CH
-
Estimate the H values for each of the following gas-phase reactions using bond dissociation energies. H2C===CH2 + Cl2 + CI-CH2CH2-C
-
Factory overhead of $44,700 consists of Indirect labor of $21,900, Depreciation expense-Factory of $16,900, and Factory utilities of $5,900. a. Compute total manufacturing costs. b. Prepare a...
-
In Giannakakos et al. (2016), titled Using Video Modeling with voiceover Instruction plus feedback to train staff to implement direct teaching procedures, why do you think collecting data on the...
-
Presidio, Inc., produces one model of mountain bike. Partial information for the company follows: Required: 1. Complete Presidios cost data table. 2. Calculate Presidios contribution margin ratio and...

Study smarter with the SolutionInn App


