A first-order decomposition reaction is observed to have the following rate constants at the indicated temperatures. Estimate
Question:
A first-order decomposition reaction is observed to have the following rate constants at the indicated temperatures. Estimate the activation energy.
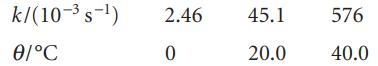
Transcribed Image Text:
k/(10-s-) Ꮎ/°C 2.46 0 45.1 20.0 576 40.0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
we have K AeEaRT so lnK2K1 E...View the full answer

Answered By

Marvine Ekina
Marvine Ekina
Dedicated and experienced Academic Tutor with a proven track record for helping students to improve their academic performance. Adept at evaluating students and creating learning plans based on their strengths and weaknesses. Bringing forth a devotion to education and helping others to achieve their academic and life goals.
PERSONAL INFORMATION
Address: , ,
Nationality:
Driving License:
Hobbies: reading
SKILLS
????? Problem Solving Skills
????? Predictive Modeling
????? Customer Service Skills
????? Creative Problem Solving Skills
????? Strong Analytical Skills
????? Project Management Skills
????? Multitasking Skills
????? Leadership Skills
????? Curriculum Development
????? Excellent Communication Skills
????? SAT Prep
????? Knowledge of Educational Philosophies
????? Informal and Formal Assessments
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A first -order decomposition reaction is observed to have the following rate constants at the indicated temperatures. Estimate the activation energy. k/(10-3 s-1) 2.46 45.1 576 0/C 20.0 40.0
-
A charged particle is observed to have a total energy of 0.638 MeV when it is moving at 0.600c. If this particle enters a linear accelerator and its speed is increased to 0.980c, what is the new...
-
A 1.00-kg object is observed to have an acceleration of 10.0 m/s2 in a direction 30.0° north of east (Fig. P5.23). The force F2 acting on the object has a magnitude of 5.00 N and is directed...
-
Harry Bhel carries a business as a sole proprietorship. During its 2022 fiscal period, its first year of operations, the business had cash sales of $123,000. It also has sales on account of $46,000,...
-
A company produces scooters used by small businesses, such as pizza parlors, that find them convenient for making short deliveries. The company is notified whenever a scooter breaks down, and the...
-
A copper pipe has an inside diameter of 3.00cm and an outside diameter of 5.00cm (Fig. 18-37). What is the resistance of 10.0m of this pipe? 3.00 cm 5.00 cm IF
-
Construct an environmental profile for a company of your choice. How might the profile be of use to management?
-
The band brake shown is used to control the speed of a rotating drum. Determine the magnitude of the couple being applied to the drum knowing that the coefficient of kinetic friction between the belt...
-
Example 2 Find the slope of a secant line joining the points that corresponds to x = -2 and x = 4 on the graph of the function y = x. 1 2 .) anioq edi grilioni 10 prizloz vd rare
-
Consider a bagel store selling three types of bagels that are produced according to the process flow diagram outlined below. We assume the demand is 180 bagels a day, of which there are 30 grilled...
-
The rate law for the reaction in Exercise 22.2a was reported as d[C]/dt = k[A][B][C]. Express the rate law in terms of the reaction rate; what are the units for k in each case? Data in Exercise 22.2a...
-
The gas-phase decomposition of acetic acid at 1189 K proceeds by way of two parallel reactions: What is the maximum percentage yield of the ketene CH 2 CO obtainable at this temperature? (1) CH3COOH...
-
Let A be the matrix given in Example 2. Show that (1, 2, 1)t, (1,1, 1)t, and (1, 0, 1)t are eigenvectors of AtA corresponding to, respectively, the eigenvalues 1 = 5, 2 = 2 and 3 = 1.
-
"A bottle of water can be 50 cents at a supermarket. $2 at the gym, $3 at the movies and $6 on a plane. Same water! Only thing that changed its value was the place." Please use what you've learned in...
-
Given functions f(x) = notation. f(x) Domain of : g(x) and g(x) = x-4, state the domains of the following functions using interval Domain of f(g(x)) (-00,-2) u (2,00) Domain of g(f(x)) (0,0)
-
Prepare the user documentation using the flowcharts created. User documentation should include an overview of menus and data entry screens, contents, options, and processing instructions. Review the...
-
How do advances in process intensification and microfluidic technologies enable the development of compact and modular separation systems with reduced footprint and improved efficiency, suitable for...
-
What are some strategies that can be used to or potential success for a company like Yogo game that originally started in a different country, using a SWOT analysis to make sure that this company can...
-
Here is some information about Stoke church Inc.: Beta of common stock = 1.2 Treasury bill rate = 4% Market risk premium = 7.5% Yield to maturity on long-term debt = 6% Book value of equity = $440...
-
Marc Company assembles products from a group of interconnecting parts. The company produces some of the parts and buys some from outside vendors. The vendor for Part X has just increased its price by...
-
Discuss the features, advantages, and limitations of the Michaelis-Menten mechanism of enzyme action.
-
Distinguish between the primary quantum yield and overall quantum yield of a chemical reaction. Describe an experimental procedure for the determination of the quantum yield.
-
Summarize the main features of the Forster theory of resonance energy transfer. Then, discuss FRET in terms of Forster theory.
-
Employee longevity A large insurance company has developed a model to identify the factors associated with employee turnover. The dependent variable is number of years an employee stays with the...
-
5. Consider the 'top-hat' function which is zero everywhere except between -1 and 1 where it takes value 1. Show, using only elementary integration, that the convolution of this function with itself...
-
HERE IS THE SITUATION. With attendance plummeting by 50 percent in the past decade, a new, younger executive director was hired to reverse the trend. By understanding the importance of integrating...

Study smarter with the SolutionInn App


