The gas-phase decomposition of acetic acid at 1189 K proceeds by way of two parallel reactions: What
Question:
The gas-phase decomposition of acetic acid at 1189 K proceeds by way of two parallel reactions:
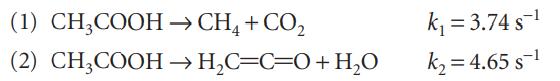
What is the maximum percentage yield of the ketene CH2CO obtainable at this temperature?
Transcribed Image Text:
(1) CH3COOH (2) CH3COOH → CH4 + CO₂ → H₂C=C=O + H₂O k₁ = 3.74 s ¹ k₂=4.65 s-¹
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 25% (4 reviews)

Answered By

Anurag Agrawal
I am a highly enthusiastic person who likes to explain concepts in simplified language. Be it in my job role as a manager of 4 people or when I used to take classes for specially able kids at our university. I did this continuously for 3 years and my god, that was so fulfilling. Sometimes I've skipped my own classes just to teach these kids and help them get their fair share of opportunities, which they would have missed out on. This was the key driver for me during that time. But since I've joined my job I wasn't able to make time for my passion of teaching due to hectic schedules. But now I've made a commitment to teach for at least an hour a day.
I am highly proficient in school level math and science and reasonably good for college level. In addition to this I am especially interested in courses related to finance and economics. In quest to learn I recently gave the CFA level 1 in Dec 19, hopefully I'll clear it. Finger's crossed :)
4.80+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Estimate the liquid diffusivity of acetic acid at 25oC in a dilute solution of:(a) Benzene,(b) Acetone,(c) Ethyl acetate, and(d) Water by an appropriate method. Compare the estimated values with the...
-
Find the viscosity of Acetic acid at 300K.
-
Calculate the volume of 47 g of acetic acid at 20 C ( = 1.05 g / mL).
-
How do recruitment and selection practices contribute to high performance in an organization?
-
PricewaterhouseCoopers Saratoga, in its 2005/2006 Human Capital Index Report, indicated the average number of days it took for an American company to fill a job vacancy in 2004 was 48 days. Sample...
-
A tungsten filament used in a flashlight bulb operates at 0.20 A and 3.2 V. If its resistance at 20oC is 1.5, what is the temperature of the filament when the flashlight is on?
-
Distinguish between the main approaches to impact assessment. Explain the three steps that lead to construction of an EIA index. Can environmental impact added ever be negative?
-
To study damage to aircraft that collide with large birds, you design a test gun that will accelerate chicken-sized objects so that their displacement along the gun barrel is given by x = (9.0 X 103...
-
Activity Assembly Product design Electricity Setup Total Budgeted Cost $ 477,750 73,500 24,500 Activity Cost Driver Direct labor hours (DLH) Engineering hours (EH) Machine hours (MH) Budgeted...
-
Use PSpice to determine Vo in the circuit of Fig. 10.124. Assume Ï = 1 rad/s. :/2 25 30 10 4709 A
-
A first-order decomposition reaction is observed to have the following rate constants at the indicated temperatures. Estimate the activation energy. k/(10-s-) /C 2.46 0 45.1 20.0 576 40.0
-
The following data have been obtained for the decomposition of N 2 O 5 (g) at 67C according to the reaction 2 N 2 O 5 (g) 4 NO 2 (g) + O 2 (g). Determine the order of the reaction, the rate...
-
Compute the condition number for using the Euclidean norm. 1 1 12 13 0 1 1 2
-
Suppose f(x) = 2x - 10x + 8. Compute the following: === A.) f(-3)+f(5) = B.) f(3) f(5) = - I
-
Month Maintenance Costs Machine Hours Health Insurance Number of Employees Shipping Costs Units Shipped January $ 4 , 5 1 0 1 7 0 $ 8 , 5 7 0 6 3 $ 2 8 , 2 4 0 7 , 0 6 0 February $ 4 , 4 7 3 1 1 0 $...
-
For individuals, economists typically identify MONEY and TIME as the two key sources of scarcity. When it comes to our standard of living, we typically focus on money in particular. Our utility is...
-
Please discuss the representation of a particular minority or disadvantaged group (or a group that has historically been marginalized) in a particular communication technology. How was that group...
-
How do the principles of process integration and energy conservation drive the development of innovative separation processes, such as reactive distillation, extractive distillation, and membrane...
-
Reliable Electric is a regulated public utility, and it is expected to provide steady dividend growth of 5% per year for the indefinite future. Its last dividend was $5 per share; the stock sold for...
-
Outline some of the major problems confronting an international advertiser.
-
On the basis of the following proposed mechanism, account for the experimental fact that the rate law for the decomposition 2 N2O5 (g) → 4 NO2 (g) + O2 (g) is v=k [N205]' k,ki (I) NO NO.+NO, (2)...
-
Consider the following mechanism for the thermal decomposition ofR2: Where R2, PA' PB are stable hydrocarbons and Rand R' are radicals. Find the dependence of the rate of decomposition of R, on the...
-
Refer to Fig. 23.3 and determine the pressure range for a branching chain explosion in the hydrogen-oxygen reaction at (a) 700 K, (b) 900 K.
-
Find a linear equation for the line that passes through (7,5) and (3,17)
-
Your firm has just been retained by Harley Quinn (38 years old) for a divorce and custody case. She and John Joker (42 Years Old) have been married for 10 years. She is currently residing in the...
-
Explain the difference between a "SELECT" and an "INSERT" SQL statement.

Study smarter with the SolutionInn App


