A simulated infrared absorption spectrum of a gas-phase organic compound is shown in the following figure. Use
Question:
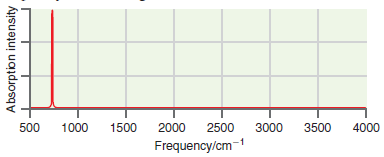
Transcribed Image Text:
500 1000 1500 2000 2500 3000 3500 4000 Frequency/cm-1 Absorption intensity
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
The single peak near 700 cm ...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A simulated infrared absorption spectrum of a gas phase organic compound is shown in the following figure. Use the characteristic group frequencies listed in Section 19.5 to decide whether this...
-
An infrared absorption spectrum of an organic compound is shown in the following figure. Use the characteristic group frequencies listed in Section 19.5 to decide whether this compound is more likely...
-
An infrared absorption spectrum of an organic compound is shown in the following figure. Use the characteristic group frequencies listed in Section 19.5 to decide whether this compound is more likely...
-
F udy A big part of communication is sharing personal information with another person. Some information we believe we have the right to be our own and should remain private. However, the degree to...
-
Rewrite tan 3x in terms of tan x.
-
What is the difficulty in applying Gauss elimination to the following linear algebraic equations? \[\begin{aligned}-10 x_{1}+10 x_{2} & =10 \\5 x_{1}-5 x_{2} & =-10\end{aligned}\]
-
a. What authorizations are necessary in executing purchases transactions? b. What errors and irregularities may occur when these transactions are not authorized?
-
The expected annual returns are 15% for investment 1 and 12% for investment 2. The standard deviation of the first investments return is 10%; the second investments return has a standard deviation of...
-
what are your thoughts on the process in conducting layoffs and the potential effect of the overall capability of an organization? from an hr strategy standpoint, how can actions be taken to help...
-
Market research has produced the following estimates of cross elasticity of demand. Branded cola and branded iced tea ............................... +1.5 Branded cola and own label cola...
-
The molecules 16 O 12 C 32 S and 16 O 12 C 34 S have values for h/8Ï 2 I of 6081.490 Ã 10 6 s 1 and 5932.816 Ã 10 6 s 1 , respectively. Calculate the CO and CS bond distances. The...
-
The rotational constant for 14 N 2 determined from microwave spectroscopy is 1.99824 cm 1 . Calculate the bond length in 14 N 2 to the maximum number of significant figures consistent with this...
-
A cylindrical specimen of cold-worked steel has a Brinell hardness of 240. (a) Estimate its ductility in percent elongation. (b) If the specimen remained cylindrical during deformation and its...
-
BuyCo, Incorporated, holds 2 5 percent of the outstanding shares of Marqueen company and appropriately applies the equity method of accounting. Excess cost amortization ( related to a patent )...
-
Wages of $11,000 are earned by workers but not paid as of December 31. Depreciation on the company's equipment for the year is $11,440 The Supplies account had a $360 debit balance at the beginning...
-
Burrell Company purchased a machine for $ 3 5 , 0 0 0 on January 2 , Year 1 . The machine has an estimated service life of 5 years and a zero estimated residual value. The asset earns income before...
-
Sierra Company manufactures soccer balls in two sequential processes: Cutting and Stitching. All direct materials enter production at the beginning of the cutting process. The following information...
-
On May 1 , 2 0 2 4 , Varga Tech Services signed a $ 1 0 8 , 0 0 0 consulting contract with Shaffer Holdings. The contract requires Varga to provide computer technology support services whenever...
-
Use a graphing utility to find the derivative of each function at the given number. f(x) = x sinx at /3
-
Tarick Toys Company manufactures video game consoles and accounts for product costs using process costing. The following information is available regarding its June inventories. The following...
-
A solution is prepared by dissolving 114 g of glucose (C 6 H 12 O 6 ) in 0.500 kg of water. The final volume of the solution is 590 mL. Calculate each value for this solution: a) molarity b) morality...
-
A solution is prepared from 445 g of ethylene glycol (C 2 H 6 O 2 ) and 500 g of water. This solution represents one that is 50% by volume ethylene glycol. At what temperature will the water in this...
-
A solution contains 0.481 mol of Na 2 SO 4 and 10.0 mol water. Calculate the vapor pressure of the solution at 25C. The vapor pressure of pure water at 25 C is 23.8 torr.
-
Employee longevity A large insurance company has developed a model to identify the factors associated with employee turnover. The dependent variable is number of years an employee stays with the...
-
5. Consider the 'top-hat' function which is zero everywhere except between -1 and 1 where it takes value 1. Show, using only elementary integration, that the convolution of this function with itself...
-
HERE IS THE SITUATION. With attendance plummeting by 50 percent in the past decade, a new, younger executive director was hired to reverse the trend. By understanding the importance of integrating...

Study smarter with the SolutionInn App


