An infrared absorption spectrum of an organic compound is shown in the following figure. Use the characteristic
Question:
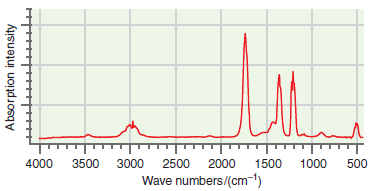
Transcribed Image Text:
T. 4000 3500 3000 2500 2000 1500 1000 500 Wave numbers/(cm-1) Absorption intensity
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
The major peak near 1700 cm 1 is the CO stretch and the peak nea...View the full answer

Answered By

Grace Igiamoh-Livingwater
I am a qualified statistics lecturer and researcher with an excellent interpersonal writing and communication skills. I have seven years tutoring and lecturing experience in statistics. I am an expert in the use of computer software tools and statistical packages like Microsoft Office Word, Advanced Excel, SQL, Power Point, SPSS, STATA and Epi-Info.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The market for oil-based paint is shown in the following table: Suppose the production of the paint creates a negative externality of $10 for each unit of paint, which is the cost of repairing the...
-
Use the following information to decide whether this equipment lease qualifies as an operating, sales-type, or direct financing lease to a lessor. (a) There is no transfer of ownership at the end of...
-
The part shown in the following figure is a carbon-steel segment (partial) gear. The smaller hole at the bottom is for clamping the part onto a round shaft, using a screw and a nut. Suggest a...
-
An important U.S. government organization charged with setting human resource management guidelines is O the EEOC (Equal Employment Opportunity Commission). the OSHA (Occupational Safety and Health...
-
In Exercises 1-2, use the given conditions to find the exact values of sin 2u, cos 2u, and tan 2u using the double-angle formulas. 1. sin u = 3 / 5, 3 / 2 < u < 2 2. cos u = 4 / 5, / 2 < u <
-
Consider the hypotheses H0 : 0 H1 : 0 where 2 is known. Derive a general expression for determining the sample size for detecting a true mean of 1 0 with probability 1 if the type I error is .
-
The probability distribution for the personality inventory test for passive aggressive traits discussed in Example 2 is shown below. Find the mean score. Data from Example 2 An industrial...
-
Accounting for securities available for sale. Events related to Simmons Corporation's investments of temporarily excess cash appear below. The firm classifies these investments as securities...
-
explain the significance of partition coefficients in determining the distribution of solutes between immiscible phases, and how does this concept guide the choice of solvents in extraction processes...
-
The Adventure Toys Company manufactures a popular line of action figures and distributes them to toy stores at the wholesale price of $10 per unit. Demand for the action figures is seasonal, with the...
-
The rotational constant for 14 N 2 determined from microwave spectroscopy is 1.99824 cm 1 . Calculate the bond length in 14 N 2 to the maximum number of significant figures consistent with this...
-
Calculate the zero point energies for 1 H 19 F and 2 D 19 F. Compare the difference in the zero point energies to k B T at 298 K.
-
Selected comparative financial statements of Haroun Company follow. Required 1. Compute trend percents for all components of both statements using 2015 as the base year. Round percents to one...
-
Particle 1 carrying charge \(4 q\) is fixed at the origin of an \(x y\) coordinate system, and particle 2 carrying charge \(q\) is fixed at \((0,0.12 \mathrm{~m})\). Particle 3 carrying \(2.0 \mu...
-
You just learned about the ethical considerations of business. Consider how these ideas relate to some of the demands business owners face. What additional information would an owner need to create...
-
The practice of corporate philanthropy a. is only a marketing strategy. b. can build stronger relationships with employees and the community at large. c. states that success can be found by competing...
-
Which of the following strategies is not designed to help a company recover from an ethical lapse? a. Ensuring whistle-blowers face legal consequences b. Working to find a leader who will set an...
-
For the high gas velocity used assume that film diffusion does not offer any resistance to transfer and reaction. Reaction temperature = 900C. Assuming that reaction proceeds by the shrinking-core...
-
Evaluate the derivatives of the following functions. f(x) = sin (tan -1 (ln x))
-
What are some of the features of the Unified Process (UP)?
-
Consider the reaction: 2 COF 2 (g) CO 2 (g) + CF 4 (g) Kc = 2.00 In an equilibrium mixture, the concentration of COF 2 is 0.35 M and the concentration of CO 2 is 0.144 M. What is the equilibrium...
-
Consider the reaction: N 2 O 4 (g) 2 NO 2 (g) Kc = 0.36 A reaction mixture initially contains [N 2 O 4 ] = 0.100 M. Find the equilibrium concentrations of N 2 O 4 and NO 2 .
-
Consider the reaction: 2 H 2 S (g) 2 H 2 (g) + S 2 (g) Kc = 1.67 10 -7 A reaction mixture initially contains [H 2 S] = 0.010 M. Find the equilibrium concentrations of H 2 and S 2 .
-
1. What year are you preparing your students for? 1973? 1995? Provide evidence that your school's curriculum and the program you use are preparing your students for 2030? Are you even preparing them...
-
2 Classify each type of numerical data as discrete (D) or continuous (C). a number of homes with solar power b patients' body temperatures c sizes of the crowds at rock concerts d star ratings of...
-
What role does the use of either mimesis or abstraction play in each example? (Keep in mind that the concepts of mimesis and abstraction in the creative arts are defined and explored in the document...

Study smarter with the SolutionInn App


