A simulated infrared absorption spectrum of a gas-phase organic compound is shown in the following figure. Use
Question:
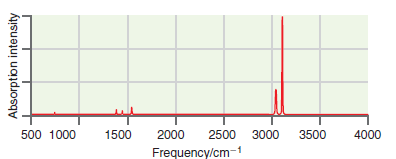
Transcribed Image Text:
500 1000 1500 2000 4000 2500 3000 3500 Frequency/cm-1 Absorption intensity
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
The peaks near 3100 cm 1 are indicative of CH stre...View the full answer

Answered By

Leah Muchiri
I am graduate in Bachelor of Actuarial Science and a certified accountant. I am also a prolific writer with six years experience in academic writing. My working principle are being timely and delivering 100% plagiarized free work. I usually present a precised solution to every work am assigned to do. Most of my student earn A++ GRADE using my precised and correct solutions.
4.90+
52+ Reviews
125+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A simulated infrared absorption spectrum of a gas phase organic compound is shown in the following figure. Use the characteristic group frequencies listed in Section 19.5 to decide whether this...
-
The investment shown in the following figure has an annual equivalent worth of $200 at i = 8%. Determine the cash flows in periods 2, 3, 5 and 6. $400 $400 3 4 5 6. Years $2,145
-
A tensile testing machine is shown in the following figure to determine the behavior of materials as they are deformed. (a) Use the provided data to calculate the stress and the corresponding strain...
-
A charge q is placed a distance from the origin, and a charge 2q is placed a distance 2r. There is a charge Q at the origin. If a charges are positive, which charge is at the higher potential? A q B)...
-
The equation of a standing wave is obtained by adding the displacements of two waves traveling in opposite directions (see figure). Assume that each of the waves has amplitude A, period T, and...
-
Is an oscillating object in translational equilibrium?
-
The price of a share is \(\$ 40\). If \(\mu=0.1\) and \(\sigma^{2}=0.16\) per year, find a \(95 \%\) confidence interval for the price of the share after six months (i.e. an interval...
-
Vail Resorts, Inc., owns and operates five premier year-round ski resort properties (Vail Mountain, Beaver Creek Resort, Breckenridge Mountain, and Keystone Resort, all located in the Colorado Rocky...
-
Create a statement of cash flow for the current year using Wright Co's income statement and balance sheet. (Do not round intermediate calculations. Round your answer to 2 decimal places.) Income...
-
The unadjusted trial balance of Fashion Centre Ltd. contained the following accounts at November 30, the company's fiscal year end: Additional information and adjustment data: 1. The 12-month...
-
Fill in the missing step in the derivation that led to the calculation of the spectral line shape in Figure 19.24. Starting from and neglecting the first term in the parentheses, show that Figure...
-
Isotopic substitution is used to identify characteristic groups in an unknown compound using vibrational spectroscopy. Consider the C~C bond in ethane ( 12 C 2 1 H 6 ). By what factor would the...
-
A couple plans to have five children. Find the probability that they will have more than three girls.
-
Paul Company has three product lines in its retail stores: books, videos, and music. The allocated fixed costs are based on units sold and are unavoidable. Demand of individual products is not...
-
Manal Company requires three units of P1 for every unit of A15 that it produces. Currently, P1 is made by Manal, with the following per unit costs in a month when 4,000 units were produced: Variable...
-
What are labour productivity and potential GDP in 2017? The first table describes an economys labour market in 2017 and the second table describes its production function in 2017. Real wage rate...
-
What are the equilibrium real wage rate and the quantity of labour employed in 2017? The first table describes an economys labour market in 2017 and the second table describes its production function...
-
Distinguish between MBO and BARS as appraisal strategies.
-
In problem, solve each equation. Express irrational solutions in exact form and as a decimal rounded to 3 decimal places. 9 2x = 27 3x-4
-
Using the information presented in Problem 13.4B, prepare a partial statement of cash flows for the current year, showing the computation of net cash flows from operating activities using the...
-
Before a rotameter can be used to measure an unknown flow rate, a calibration curve of flow rate versus rotameter reading must be prepared. A calibration technique for liquids is illustrated below. A...
-
How many of the following are found in 15.0 kmol of xylene (C 8 H 10 )? (a) kg C 8 H 1 ; (b) mol C 8 H 1 ; (c) lb-mole C 8 H 1 ; (d) mol (g-atom) C; (e) mol H; (f) g C; (g) g H; (h) molecules of C 8...
-
Certain solid substances, known as hydrated compounds, have well-defined molecular ratios of water to some other species. For example, calcium sulfate dihydrate (commonly known as gypsum, (CaSO 4 2H...
-
4. Given the weighted graph: A B 9 F 5 4 9 6 E D Execute Prim's minimum spanning tree algorithm starting with vertex G. List the edges and their weights in the order they are added to the tree. Also,...
-
1. Given the directed graph: B D G Perform a depth-first search starting with vertex G, assuming an adjacency list is in alphabetical order for each vertex. List the vertices in the order they are...
-
Write the following if statements in C, in Assembly language using a) subtract/carrier-flag test and b) comparisons instructions. 1) Write the following statement in C, in Assembly language (10...

Study smarter with the SolutionInn App


