A simulated infrared absorption spectrum of a gas-phase organic compound is shown in the following figure. Use
Question:
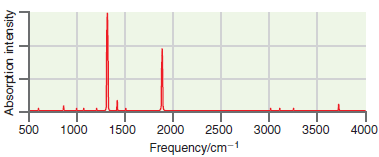
Transcribed Image Text:
2000 500 1000 1500 2500 3000 3500 4000 Frequency/cm-1 Absorption intensity
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (15 reviews)
The peak near 3700 cm 1 is indicative of an OH s...View the full answer

Answered By

Sumit kumar
I am an experienced online essay writer with a thorough understanding of any curriculum.and subject expert at Chegg for mathematics, CS subjects..
4.90+
5+ Reviews
13+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A simulated infrared absorption spectrum of a gas phase organic compound is shown in the following figure. Use the characteristic group frequencies listed in Section 19.5 to decide whether this...
-
An infrared absorption spectrum of an organic compound is shown in the following figure. Use the characteristic group frequencies listed in Section 19.5 to decide whether this compound is more likely...
-
An infrared absorption spectrum of an organic compound is shown in the following figure. Use the characteristic group frequencies listed in Section 19.5 to decide whether this compound is more likely...
-
A credit card contains 16 digits. It also contains a month and year of expiration. Suppose there are one million users of a credit card with unique card numbers. A hacker randomly selects a 16 digit...
-
Consider the function f(x) = cos 1 / x and its graph, shown in the figure below. (a) What is the domain of the function? (b) Identify any symmetry and any asymptotes of the graph. (c) Describe the...
-
What are econometric foreign exchange forecasting models?
-
Carlson Corporation accountants have assembled the company's data for the year ended December 31, 2007. Requirement Prepare Carlson Corporation's statement of cash flows using the indirect method to...
-
Following are the four assertions about account balances that can be applied to the audit of a companys PP& E, including assets the company has constructed itself: existence, rights and obligations,...
-
TLC company expects constant 3% growth in a market that expects 6% on investments. ABC currently sells for $91.00 per share. What would you expect their next dividend to be?
-
The Carlson Department Store suffered heavy damage when a hurricane struck on August 31, 2003. The store was closed for four months (September 2003 through December 2003), and Carlson is now involved...
-
The rotational energy of 7 Li 2 in the J = 5 state is 4.0126 10 22 J. Calculate the bond length of the molecule.
-
If the vibrational potential is not harmonic, the force constant is not independent of degree of stretching or compression of a molecule. Using the relation k effective = (d 2 V(x)/dx 2 ), derive an...
-
Consider a chloroform(1)/tetrahydrofuran(2) mixture with x 1 = 0.90, initially at 76C and 120 kPa. Describe the evolution of phases and phase compositions as the temperature is gradually reduced to...
-
1. Consider a point moving at a constant speed v on a circle of radius r. What is the relation between v, r, and the rotation frequency, f? 2. Show that the centripetal acceleration relation, ac =...
-
A small bead of mass, m, slides freely inside a smooth tube with length 2L. The tube is spun about its center with constant angular speed, \theta\dot. Imagine that with the tube spinning, the bead is...
-
The 2,842-seat performing arts center has three sections-orchestra, mezzanine, and balcony. The mezzanine has one-sixth fewer seats than the orchestra. How many seats are in the balcony if the...
-
You invest $920 at the end of each six months for the next 13 years. What is the value of your investment at the end of 13 years if your investment earns 5.25% compounded semi-annually?
-
A sample of 100 clients of an exercise facility was selected. Let X = the number of days per week that a randomly selected client uses the exercise facility. X Frequency 03 1 13 2 30 3 28 4 11 5 9 6...
-
Are the parties to a contractual joint venture, a strategic alliance, a distributorship, and a franchise agreement in an agency relationship?
-
Which of the following statements is false? a. Capital leases are not commonly reported in a Capital Projects Fund. b. A governmental entity may report a Capital Project Fund in one year but not the...
-
In a metered-dose inhaler (MDI), such as those used for asthma medication, medicine is delivered by a compressed-gas propellant. (The device is similar in concept to a can of spray paint.) When the...
-
A stream of propane at an average temperature T = 566R and absolute pressure P = 6.8 atm flows from a hydrocarbon processing plant to a nearby customers production facility. A technician at the...
-
A 150-liter cylinder of carbon monoxide is stored in a 30.7-m 3 room. The pressure gauge on the tank reads 2500 psi when the tank is delivered. Sixty hours later the gauge reads 2245 psi. The...
-
A physiotherapist purchased 7 rolls of resistance bands and 12 therapy balls for a total cost of $406. A second purchase, at the same prices, included 9 rolls of resistance bands and 14 therapy...
-
The Mentor company manufactures butcher block tables. Each table requires the following direct materials, direct labor and overhead. Standard Quantity 2" clear maple 12 db. Ft. Direct labor 2 hours...
-
Vintage is a public company that prepares financial statements inaccordance with IFRS Standards. Its reporting date is 31 March 20X3. Vintage entered into a contract with a customer to supply...

Study smarter with the SolutionInn App


