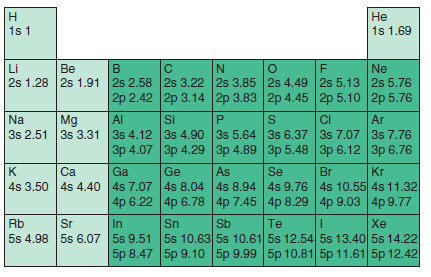
Are the effective nuclear charges listed in Figure 21.13 helpful in explaining the trend in the electron
Question:
Transcribed Image Text:
Н Не 1s 1 1s 1.69 Li 2s 1.28 2s 1.91 Be Ne 2s 2.58 2s 3.22 2s 3.85 2s 4.49 2s 5.13 2s 5.76 2p 2.42 2p 3.14 2p 3.83 2p 4.45 2p 5.10 2p 5.76 Mg Si 3s 2.51 3s 3.31 3s 4.12 3s 4.90 3s 5.64 3s 6.37 3s 7.07 3s 7.76 | Зр 4.07 | Зр 4.29 |Зр 4.89 | Al CI Na Ar Зр 5.48 |Зр 6.12 | Зр 6.76 Se Ca Ga Ge As Br Kr 4s 3.50 4s 4.40 4s 7.07 4s 8.04 4s 8.94 4s 9.76 4s 10.55 4s 11.32 4p 6.22 4p 6.78 4p 7.45 4p 8.29 4p 9.03 4p 9.77 Sn 5s 4.98 5s 6.07 5s 9.51 5s 10.63 5s 10.61 5s 12.54 5s 13.40 5s 14.22 Rb Sr In Sb Te Xe 5p 8.47 5p 9.10 5p 9.99 5p 10.81 5p 11.61 5p 12.42
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
They are less useful than in understanding the tr...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A. Give a name to match the structure for the following compound. CH3 COOH Spell out the full name of the compound. Submit Request Answer Part B Give a name to match the structure for the following...
-
Explain why the first electron affinity of sulfur is 200 kJ/mol but the second electron affinity is - 649 kJ/mol.
-
Explain why the electron affinity of nitrogen is approximately zero, while the elements on either side, carbon and oxygen, have substantial positive electron affinities.
-
All of the following represent trends shaping HR over the past decade, according to our reading, except: Organizations are spending more on the expansion of physical office space Employees are...
-
Find the exact value of the expression. 1. cos (arctan 3 / 4) 2. tan (arcos 3 / 5) 3. sec (arctan 12 / 5) 4. cot [ arcsin (12 / 13)]
-
Consider the balance-of-payments accounting information for FarawayLand as shown in the table below for the year 2018. All values are in billions of dollars. a. What is the trade balance? b. What is...
-
If you were asked to develop a training plan for an HRIS implementation, what kinds of things would you include? Why?
-
Harris owned a farm that was worth about $600 per acre. By false representations of fact, Harris induced Pringle to buy the farm at $1,500 per acre. Shortly after taking possession of the farm,...
-
Mark opens his own company and completes the following transactions in May: 5/1 Mark invests $14,000 into the business. 5/3 Purchased $3,800 of equipment on account. 5/4 Paid $560 premium for a...
-
Earl sells lemonade in a competitive market on a busy street corner in Philadelphia. His production function is f(x1, x2) = x11/3 x21/3, where output is measured in gallons, x1 is the number of...
-
Explain why the electron affinity of N is negative.
-
Are the effective nuclear charges listed in Figure 21.13 helpful in explaining the trend in the first ionization energy with increasing atomic number? Explain your answer. 1s 1 1s 1.69 Li 2s 1.28...
-
In industry, performance based logistics (PBL) strategies are increasingly popular ways to reduce cost, increase revenue, and attain customer satisfaction. The Journal of Business Logistics (Vol. 36,...
-
In a week, Erin buys 1 six-pack of soda and sees 2 movies when a movie ticket is $10, soda is $5 a six-pack, and she has $25 to spend. If her budget increases and she has $50 to spend on soda and...
-
Novartis International AG, a global healthcare company based in Switzerland, is considering producing either a generic multivitamin or a specific blend of vitamins A, C, and D. Novartis expects both...
-
The Age Discrimination in Employment Act prohibits discrimination against persons________________ a. 40 years and older b. 50 years and older c. 65 years and older d. of any age
-
The system of dual reporting relationships-to both a functional and project manager-is characteristics of a structure. a. functional b. matrix c. network d. product
-
List two symptoms that would alert a manager that a team is suffering from groupthink. What could this manager do to counteract each of these symptoms?
-
Gravity on Earth If a rock falls from a height of 100 meters on Earth, the height H (in meters) after t seconds is approximately H(t) = 100 - 4.9t 2 (a) In general, quadratic functions are not...
-
(a) With not more than 300 words, examine why Malaysia missed its 2020 targets to divert 40% of waste from landfills and increase recycling rates to 22%. (10 marks) b) Why arent Malaysian recycling?...
-
Haemoglobin, the red blood protein responsible for oxygen transport, binds about 1.34 cm 3 of oxygen per gram. Normal blood has a haemoglobin concentration of 150 g dm 3 . Haemoglobin in the lungs is...
-
K. Sato, F.R. Eirich, and J.E. Mark (J. Polymer Sci., Polym. Phys. 14, 619 (1976)) have reported the data in the table below for the osmotic pressures of polychloroprene ( = 1.25 g cm 3 ) in toluene...
-
The mean activity coefficients of HBr in three dilute aqueous solutions at 25C are 0.930 (at 5.0 mmol kg 1 ), 0.907 (at 10.0 mmol kg 1 ), and 0.879 (at 20.0 mmol kg 1 ). Estimate the value of B in...
-
4. Methane burns in air with an equivalence ratio of =0.85, determine the composition of the products: What are the Mass Fractions of each species in the Products?
-
1. Describe the health and safety requirements of the area in which the installation activity is to take place, and the responsibility these requirements place on the learner 2. Describe the...
-
1. Which is easier for nucleation in solids, homogeneous or heterogeneous nucleation? What are the common defects present in solid solution (list at least five kinds of defects)? Are the defects...

Study smarter with the SolutionInn App


