Careful measurements of the potential of the cell Pt|H 2 (g,p )|NaOH(aq, 0.0100mol kg 1 ),Nacl(aq,
Question:
Careful measurements of the potential of the cell Pt|H2(g,p⦵)|NaOH(aq, 0.0100mol kg−1),Nacl(aq, 0.011 25mol kg−1)|AgCl(s)|Ag(s) have been reported. Among the data is the following information:
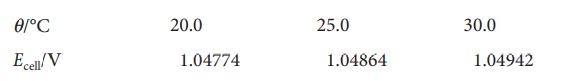
Calculate pKw at these temperatures and the standard enthalpy and entropy of the autoprotolysis of water at 25.0 °C.
Transcribed Image Text:
ᎾᏟ Ecell/V 20.0 1.04774 25.0 1.04864 30.0 1.04942
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
At 0 C the pKw can be calculated using the equation Ecell Eocell 005916nlogKw Substituting the value...View the full answer

Answered By

Dansteve Matoke
As a consequence of more than three years of experience in ACADEMIC WRITING, I have vast, diverse knowledge and impeccable grammar. I guarantee the quality of my work across multiple fields. I intend to precisely meet the clients' expectations. Let's work together to achieve exceptional grades.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Physical Chemistry Thermodynamics And Kinetics
ISBN: 9781464124518
10th Edition
Authors: Peter Atkins, Julio De Paula
Question Posted:
Students also viewed these Sciences questions
-
The temperature dependence of the potential of a cell is vanishingly small. What does this tell you about the thermodynamics of the cell reaction?
-
Measurements of the potential at points on the axis of a charged disk are given in the two tables below, one for measurements made close to the disk and the other for measurements made far away. In...
-
Recession of the Moon Careful measurements of the earth-moon separation indicate that our satellite is presently moving away from us at approximately 3.0 cm per year. Neglect any angular momentum...
-
In December 2014, Shire Computer's management establishes the 2015 predetermined overhead rate based on direct labor cost. The information used in setting this rate includes estimates that the...
-
Henne Optical Corporation reported the following information regarding long-term operating assets for its Lens Manufacturing Operations: Recent advances in technology have rendered the companys lens...
-
The balance sheet debit column of the worksheet for Shaw Company includes the following accounts: Accounts Receivable $12,500; Prepaid Insurance $3,600; Cash $4,100; Supplies $5,200 and Short-term...
-
For each of the following residual plots, determine whether the assumptions of the linear model are satisfied. If they are not, specify which assumptions are violated. b. C. d.
-
On January 1, 2018, Allied Industries leased a high-performance conveyer to Karrier Company for a four-year period ending December 31, 2021, at which time possession of the leased asset will revert...
-
At December 31, 2023, the Accounts Receivable balance of Foley Distribution Service is $356,000. The Allowance for Doubtful Accounts has a(n) $9,900 credit balance. Accountants for Foley Distribution...
-
In an air-filled rectangular waveguide, a TE mode operating at 6 GHz has Ey = 5 sin (2x/a) cos(y/b) sin (wt 12z) V/m Determine: (a) The mode of operation, (b) The cutoff frequency, (c) The intrinsic...
-
At 2257K and 1.00bar total pressure, water is 1.77 per cent dissociated at equilibrium by way of the reaction 2 H 2 O(g)2 H 2 (g)+O 2 (g). Calculate K.
-
The standard Gibbs energy of the reaction N 2 (g)+3 H 2 (g)2 NH 3 (g) is 32.9 kJmol 1 at 298K. What is the value of r G when Q=(i) 0.010, (ii) 1.0, (iii) 10.0, (iv) 100 000, (v) 1 000 000? Estimate...
-
Describe the two types of paper-and-pencil honesty tests. Should your organization make job applicants complete one? Why or why not?
-
Under what circumstances do overhead electrical lines and communications cables present a hazard when operating an MEWP?
-
From 2016 and 2017, what accounts for the high ratios of Goodwill to total assets (over 30%) for the following companies: United Healthcare, Proctor and Gamble, and Pfizer? On the other hand, why is...
-
ToyJoy! estimates that customers will be granted 2,600 in refunds of this year's sales next year and the merchandise expected to be returned will have a cost $2,000. How would I journalize the...
-
When evaluating a project, the general rule is that if the NPV is positive, you should accept the project; if it is negative, you would reject the project. From the reading this week, we also learned...
-
When his income falls from $52,000 to $38,000, Victor increases his monthly purchase of Hamburger Helper from 8 boxes to 10 boxes. From the midpoint, Victor's income elasticity of demand for...
-
Prove the identities in (3.82) and (3.83).
-
The Higher the time period of the financial security the higher the. ............... risk. O a. Maturity O b. Default and Maturity Oc. Default O d. Liquidity
-
Calculate the entropy of activation for a collision between two structureless particles at 500 K, taking M = 78 g mol 1 and = 0.62 nm 2 .
-
Estimate the magnitude of the corrosion current for a patch of zinc of area 0.25 cm 2 in contact with a similar area of iron in an aqueous environment at 25C. Take the exchange current densities as 1...
-
Calculate the thermodynamic limit to the zero-current potential of fuel cells operating on (a) Hydrogen and oxygen, (b) Methane and air, and (c) Propane and air. Use the Gibbs energy information in...
-
Calculate the missing amounts. (Loss amounts should be indicated with a minus sign and input all other amounts as positive values. Do not round intermediate calculations. Round "Gross profit ratio"...
-
Sharon was hired by the Beauty Mark Company to sell beauty products house to house in a certain town. Unknown to either Sharon or Beauty Mark, there is an ordinance required all salespersons to...
-
Many large employers and a growing number of small to mid-size employers have adopted self-funded employee health benefit plans. In one or more paragraphs, discuss the laws that apply to self-funded...

Study smarter with the SolutionInn App


