Consider the following isotope-exchange reaction: DCl (g) + HBr (g) DBr (g) + HCl (g) The amount
Question:
DCl (g) + HBr (g) ‡„ DBr (g) + HCl (g)
The amount of each species at equilibrium can be measured using proton and deuterium NMR (see Journal of Chemical Education 73 [1996]: 99). Using the spectroscopic information below, determine KP for this reaction at 298 K. For this reaction, Δε = 41 cmˆ’1, equal to the difference in zero-point energies between products versus reactants, and the ground-state electronic degeneracy is zero for all species.
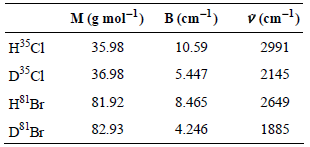
Transcribed Image Text:
M (g mol-l) в (сm-1) V (cm-1) н$C1 10.59 35.98 2991 D3°C1 36.98 5.447 2145 H$'Br 81.92 8.465 2649 D°'Br 82.93 1885 4.246
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
For DBr Notice that the vibrational partition function is approximately equal to on...View the full answer

Answered By

Mustafa olang
Please accept my enthusiastic application to solutionInn. I would love the opportunity to be a hardworking, passionate member of your tutoring program. As soon as I read the description of the program, I knew I was a well-qualified candidate for the position.
I have extensive tutoring experience in a variety of fields. I have tutored in English as well as Calculus. I have helped students learn to analyze literature, write essays, understand historical events, and graph parabolas. Your program requires that tutors be able to assist students in multiple subjects, and my experience would allow me to do just that.
You also state in your job posting that you require tutors that can work with students of all ages. As a summer camp counselor, I have experience working with preschool and kindergarten-age students. I have also tutored middle school students in reading, as well as college and high school students. Through these tutoring and counseling positions, I have learned how to best teach each age group. For example, I created songs to teach my three-year-old campers the camp rules, but I gave my college student daily quizzes to help her prepare for exams.
I am passionate about helping students improve in all academic subjects. I still remember my excitement when my calculus student received her first “A” on a quiz! I am confident that my passion and experience are the qualities you are looking for at solutionInn. Thank you so much for your time and consideration.
4.80+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The equilibrium constant Kc for the reaction is 4.2 at 1650°C. Initially 0.80 mol H2 and 0.80 mol CO2 are injected into a 5.0-L flask. Calculate the concentration of each species at equilibrium....
-
In the gas-phase reaction A + B ;:='0C + 2 D, it was found that, when 2.00 mol A, 1.00 mol B, and 3.00 mol D were mixed and allowed to come to equilibrium at 25C, the resulting mixture contained 0.79...
-
The equilibrium between hydrogen cyanide (HCN) and its isomer hydrogen isocyanide (HNC) is important in interstellar chemistry: HCN (g) HNC (g) A long-standing puzzle regarding this reaction is that...
-
Using the Trust Services Principles and Criteria for the Online Privacy Principle, develop an online privacy policy for Alltel Stadium that could be posted on the stadiums website for customers to...
-
1. How can you tell whether an ellipse is a circle from the equation? 2. Is the graph of x2 4y4 = 4 a hyperbola? Explain.
-
Vorticity is generated any time a fluid contacts a solid surface. The vorticity diffuses from the solid surface and is convected away by the fluid. Consider the heat transfer to a falling film...
-
Outline the project planning process based on identified resources.
-
Old Town Entertainment has two employees in 2016. Clay earns $3,600 per month and Philip, the manager, earns $10,800 per month. Neither is paid extra for working overtime. Assume the Social Security...
-
What is the present value of $100,000 to be received in 20 years? Your required rate of return is 8% per year. What is the annual interest rate required for 20,000 to grow into 30,000 in 15 years?
-
A company uses standard absorption costing to value inventory. Its fixed overhead absorption rate is $12 per labour hour and each unit of production should take four labour hours. In a recent period...
-
In Direct Measurement of the Size of the Helium Dimer by F. Luo, C. F. Geise, and W. R. Gentry [J. Chemical Physics 104 (1996): 1151], evidence for the helium dimer is presented. As one can imagine,...
-
The isotope exchange reaction for Cl 2 is as follows: 35 Cl 35 Cl + 37 Cl 37 Cl 237 Cl 35 Cl The equilibrium constant for this reaction is 4. Furthermore, the equilibrium constant for similar...
-
The graph of a quadratic function is called a(n) ___________.
-
If f(x) = e* x, find '(1).
-
How can organizations design marketing communication programs that keep pace with the rapid changes in technology? Provide two examples and explain how these methods will allow organizations to...
-
Delta Airlines is working on pricing its own flights between Detroit Metro Airport and Minneapolis-St. Paul Airport. Over years of flying the route, Delta knows that the weekly demand curve it faces...
-
8. Consider the equation 7 sinx- 4 sin 2x =-1. COS X a) Put the equation in standard quadratic trigonometric equation form.
-
Give one vision statement for watch brand. As this is are group project to introduce new product . And tell something about watches like first introduction , second vision statement and then What is...
-
Under the Canada Business Corporations Act the auditor has a responsibility to: (a) Form an opinion on the subject criteria. (b) Form an opinion on the independence of the company. (c) Form an...
-
If a force of F = 50 Ib is applied to the pads at A and C, determine the smallest dimension d required for equilibrium if the spring has an unstretched length of 1 ft. B 1 ft 1 ft F k = 15016/fr 1ft...
-
Identify the alkene that would yield the following products via ozonolysis: a. b. c. d.
-
Cyclopentanone was treated with lithium aluminum hydride followed by H 3 O + . Explain what you would look for in the IR spectrum of the product to verify that the expected reaction had occurred....
-
When 1-chlorobutane is treated with sodium hydroxide, two products are formed. Identify the two products, and explain how these products could be distinguished using IR spectroscopy.
-
Mrs. Sam Taffer was a brilliant engineer at IBM and made a lot of money from stock options. Since then, she has worked as a musician and raised her two sons. She wishes to have most of her property...
-
Miller Company ended its fiscal year on June 30, 2017. The company's adjusted trial balance as of the end of its fiscal year is shown below. MILLER COMPANY Adjusted Trial Balance June 30, 2017...
-
What depreciation method is used to write off property, plant and equipment? Regarding the triple bottom line, what information did the company disclose about environmental matters in the reports?...

Study smarter with the SolutionInn App


