Identify the alkene that would yield the following products via ozonolysis: a. b. c. d.
Question:
a.
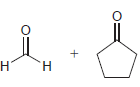
b.
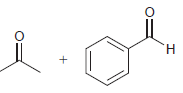
c.
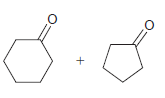
d.
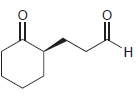
Transcribed Image Text:
Н Н
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
a...View the full answer

Answered By

Charles Okinda
students should give all the instructions concerning the challenge that they face. they will get an immediate response because I am always online.
4.90+
753+ Reviews
1481+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Give an example reaction that would yield the following products. Name the organic reactant and product in each reaction. a. Alkane b. Monohalogenated alkane c. Dihalogenated alkane d. Tetra ha...
-
Identify the alkene that would give each of the following products upon ozonolysis followed by treatment with hydrogen peroxide: a. b. c. d. e. f. CH3CH2CH2COH CH3CCH CH3CCH2CH2CH2CH2CCH2CH3 OH HO +...
-
Write the structures of the alkenes that would yield the following carbonyl compounds When treated with ozone and then with dimethyl sulfide. (a) (b) (2 Mol is produced from 1 mol of alkene) (c) o...
-
Thor Bhd. (Thor) is a listed company in Malaysia, specializes in selling batteries. At 31 December 2021, Thor holds four distinct types of batteries in its warehouse. The accountant of Thor provided...
-
Show the result of performing three deleteMin operations in the heap of the previous exercise.
-
Estimate how many hours you will spend in each of the upcoming three months doing the purchasing, repackaging, and selling. Select a reasonable wage rate for yourself. What will your total labour...
-
Three different instrument panel configurations were tested by placing airline pilots in flight simulators and testing their reaction time to simulated flight emergencies. Eight pilots were assigned...
-
After returning from a three-day business trip, the accountant for Southeast Sales, Johanna Estrada, checked bank activity in the company's checking account online. The activity for the last three...
-
Q1. Jassim Compagny is producing only one product. Two types of direct materials are used to produce this product: direct material type( A) and direct material type B. The estimated data for Jassim...
-
The position of a particle as a function of time is given by r(vector) = (5.0i + 4.0j)t 2 m, where t is in seconds. a. What is the particles distance from the origin at t = 0, 2, and 5 s? b. Find an...
-
The intermediate-term external benefits of Head Start a. Focus on reducing the likelihood of the children ending up in prison. b. Focus on reducing the likelihood that the children will become...
-
Social Securitys revenue emanates from taxes on a. All income. b. Payrolls. c. Capital. d. Estates.
-
Select the set associativity to be used by a 64 KB L1 data cache shared by both benchmarks. If the L1 cache has to be directly mapped, select the set associativity for the 1 MB L2 cache. For the...
-
The first step in cash flow statement analysis should be to: A. evaluate consistency of cash flows. B. determine operating cash flow drivers. C. identify the major sources and uses of cash.
-
The item retained earnings is a component of: A. assets. B. liabilities. C. shareholders equity.
-
Green Glory Corp., a garden supply wholesaler, reported cost of goods sold for the year of $80 million. Total assets increased by $55 million, including an increase of $5 million in inventory. Total...
-
During 2009, Accent Toys Plc., which began business in October of that year, purchased 10,000 units of a toy at a cost of 10 per unit in October. Th e toy sold well in October. In anticipation of...
-
Which of the following is an appropriate method of computing free cash flow to the firm? A. Add operating cash flows to capital expenditures and deduct after-tax interest payments. B. Add operating...
-
A stone is launched vertically upward from a cliff 192 feet above the ground at a speed of 64 ft/s. Its height above the ground t seconds after the launch is given by s = -16t 2 + 64t + 192, for 0 t...
-
Interview managers at three companies in your area about their use of ERP. How have their experiences been similar? What accounts for the similarities and differences?
-
Which of the following molecules has a dipole moment? Indicate the expected direction ofeach. (b) (d) (c) (a)
-
(a) The H C1 bond length is 136 pm. What would the dipole moment of HC1 be if the molecule were 100% ionic, H+ C1-? (b) The actual; dipole moment HC1 is 1.08 D. What is the percent ionic character...
-
Phosgene, C12C = O, has a smaller dipole moment then formaldehyde, H2C = O, even though it contains electronegative chlorine atoms in place of hydrogen. Explain
-
Draw the UML class diagram based on the following Java code. public class Propeller { public double thrust; public int mileage; public class Engine ( public double power; public int mileage; } public...
-
a) Based on the following code segment, draw the representative UML diagram. class Vehicle ( private String regNum; private String departStat; private String arriveStat; public Vehicle () ( } regNum...
-
The following linear demand specification is estimated for Conlan Enterprises, a price-setting firm: Q=a+bP+CM+dPR where Q is the quantity demanded of the product Conlan Enterprises sells, P is the...

Study smarter with the SolutionInn App


