Consider the gas-phase isomerization of cyclopropane. Are the following data of the observed rate constant as a
Question:
Are the following data of the observed rate constant as a function of pressure consistent with the Lindemann mechanism?
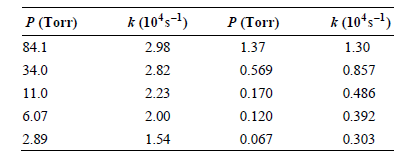
Transcribed Image Text:
CH, CH,CH=CH2 CH,-CH2 k (10ʻs-1) 2.98 2.82 P (Torr) 84.1 к (10'5-1) P (Torr) 1.37 1.30 0.569 0.170 0.857 0.486 34.0 2.23 11.0 6.07 2.00 0.120 0.392 0.067 2.89 1.54 0.303
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 87% (16 reviews)
If the data obeyed the Lindemann mechanism then a plot of the data would fit the equ...View the full answer

Answered By

Ayush Jain
Subjects in which i am expert:
Computer Science :All subjects (Eg. Networking,Database ,Operating System,Information Security,)
Programming : C. C++, Python, Java, Machine Learning,Php
Android App Development, Xamarin, VS app development
Essay Writing
Research Paper
History, Management Subjects
Mathematics :Till Graduate Level
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The isomerization of cyclopropane, C3H6, to propylene, CH2=CHCH3, is first order in cyclopropane and first order overall. At 1000oC, the rate constant is 9.2/s. What is the half-life of cyclopropane...
-
The isomerization of cyclopropane, C3H6, is believed to occur by the mechanism shown in the following equations: Here C3H6* is an excited cyclopropane molecule. At low pressure, Step 1 is much slower...
-
From the following data for the first-order gas-phase isomerization of CH3NC at 215 oC, calculate the first-order rate constant and half-life for the reaction: Time (s) Pressure CH3NC (torr)...
-
Find the exact value of sin(x - y) if sin(x) = 3T 3T T
-
Use the graph of the function f to create a table of values for the given points. Then create a second table that can be used to find f-1, and sketch the graph of f-1, if possible. 1. 2. y 4. + 2 4...
-
Suppose that X has the uniform distribution on the interval [0, 1]. Compute the variance of X.
-
The level of pollution because of vehicular emissions in a city is not regulated. Measurements by the local government of the change in flow of vehicles and the change in the level of air pollution...
-
1. Use descriptive statistics to summarize the data from the two studies. What are your preliminary observations about the depression scores? 2. Use analysis of variance on both data sets. State the...
-
Identify a major limitation in the application of a group contingency to manage behavior and give an example. You can choose an example that involves the reduction or acquisition of behavior.
-
Print-for-All is a family-owned print shop that has grown from a three-press two-color operation to a full-service facility capable of performing a range of jobs from simple copying to four-color...
-
In the discussion of the Lindemann mechanism, it was assumed that the rate of activation by collision with another reactant molecule, A, was the same as collision with a nonreactant molecule, M, such...
-
Consider the following mechanism, which results in the formation of product P: If only the species A is present at t = 0, what is the expression for the concentration of P as a function of time? You...
-
Given the significant rise in regional bond issuance following the 2008 global financial crisis, Next Europe Asset Management Limited aims to grow its assets under management by attracting a variety...
-
Given that almost each country has its own generally accepted accounting principles, why do we need a set of internationally accepted accounting standards such as IFRS?
-
What are the possible reasons for the SEC's waiver of the U.S. GAAP reconciliation for foreign registrants that followed IFRS to prepare financial statements?
-
What does IFRS stand for? Who sets up the IFRS and are there many countries adopting IFRS? What is the advantage to adopt IFRS?
-
According to the IASB, there have been more than 100 countries or jurisdictions adopting IFRS. What is the scope of IFRS when people mention this term?
-
Briefly explain the evolutionary establishment of IASB.
-
A trumpet at room temperature (25C) is tuned to play the note middle C, which has a fundamental frequency of 262 Hz. The trumpet is then taken outside, and after a few minutes, it is found that the...
-
Using the parallel-axis theorem, determine the product of inertia of the area shown with respect to the centroidal x and y axes. 6 in. 9 in. 9 in- 4.5 in. in. 4.5 in.
-
An equimolar mixture of H 2 and CO is obtained by the reaction of steam with coal. The product mixture is known as water-gas. To enhance the H 2 content, steam is mixed with water-gas and passed over...
-
For the systems specified below, obtain residue curve maps and plot the range of possible distillate and bottoms compositions for the given feeds. UNIQUAC parameters are provided. (a) methanol(1) +...
-
(a) Rank the following molecules in order of increasing oxidation of carbon and give the oxidation state of C for each: CO2, -COH(aldehyde), -COOH(carboxylic acid), -CO- (ketone), -COH(alcohol),...
-
Skysong needs to calculate the company's expected cash receipts for the upcoming month to determine whether additional financing is needed. Typically, the company's sales consist of 40% cash sales...
-
The value of NSE index is 1140. The continously compunded risk free rate is 4.6% amnd continous dividend yield is 2.1%. Calculate the no arbitrage price for a 140 day forward contract on the index
-
Explain why MNCs such as Coca Cola and PepsiCo, Inc., still have numerous opportunities for international expansion. Explain briefly

Study smarter with the SolutionInn App


