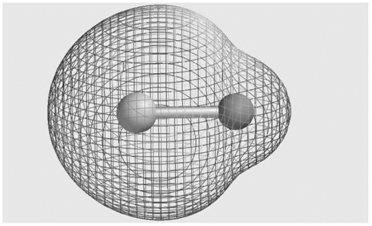
Consider the molecular electrostatic potential map for the LiH molecule shown here. Is the hydrogen atom (shown
Question:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (5 reviews)
It is an electron acceptor becaus...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the molecular electrostatic potential map for the NH 3 molecule shown here. Is the hydrogen atom (shown as a white sphere) an electron acceptor or an electron donor in this molecule?
-
Consider the molecular electrostatic potential map for the BH 3 molecule shown here. Is the hydrogen atom (shown as a white sphere) an electron acceptor or an electron donor in this molecule?
-
Consider the molecular electrostatic potential map for the BeH 2 molecule shown here. Is the hydrogen atom (shown as a white sphere) an electron acceptor or an electron donor in this molecule?
-
Workplace stress is sometimes related to time management. Describe a time when you were stressed at work or school because of time management. Explain how you would advise an employee or student to...
-
Define the inverse cosecant function by restricting the domain of the cosecant function to the intervals [/2, 0) and (0, /2], and sketch the graph of the inverse trigonometric function.
-
List and briefly explain three common strategies for the firm to finance its permanent and temporary capital needs.
-
Here's where you can apply the information in one of this chapter's tips and get a chance to predict a Super Bowl winner! Coach Kent is curious to know whether the average number of games won in a...
-
Data on the physical inventory of Rhino Company as of December 31, 2012, are presented in the working papers. The quantity of each commodity on hand has been determined and recorded on the inventory...
-
How has face book influenced political candidate's electoral success? What is the relationship between social media technology called face book and electoral success?
-
A process plant consumes of 12500 kWh per month at 0.9 power Factor (PF). What is the percentage reduction in distribution losses per month if PF is improved up to 0.96 at load end?
-
The total energy of a molecule is lowered if the orbital energy of the anti-bonding MO is negative, and raised if the orbital energy of the anti-bonding MO is positive. The zero of energy is the...
-
Consider the molecular electrostatic potential map for the H 2 O molecule shown here. Is the hydrogen atom (shown as a white sphere) an electron acceptor or an electron donor in this molecule?
-
Consider the entangled wave function for two photons, Assume that the polarization operator PË i has the properties PËÏ i i (H) = Ï i (H) and PË i Ï i (V ) = + Ï i...
-
Explain which research tradition (qualitative, quantitative, critical) appeals most to you; and how Rodriguez's (2014) positions on the colonization of the field of communication impact your selected...
-
Rearrange this expression into quadratic form, ax2 + bx + c = 0, and identify the values of a, b, and c. 0.20= b= c= 55-x
-
For the right triangle below, find the measure of the angle. Round to the hundredths. (2 decimal places) 11 14
-
The following data refer to Huron Corporation for the year 20x2. Sales revenue Raw-material inventory, 12/31/x1 Purchases of raw material in 20x2 Raw-material inventory, 12/31/x2 Direct labor cost...
-
Who are the major stakeholders in Molson Coors, and how is each group affected by the "Making History" corporate communication style that the company has chosen to use.
-
Find the sum. 7 + 1 - 5 - 11 - ... - 299
-
2.) Find the Laplace transform of f(t) 7e-St cos 2t +9 sinh2 2t. Use Laplace Table. %3D
-
What irreducible representations do the four H1s orbitals of CH 4 span? Are there s and p orbitals of the central C atom that may form molecular orbitals with them? Could d orbitals, even if they...
-
Molecules belonging to the point groups D 2h or C 3h cannot be chiral. Which elements of these groups rule out chirality?
-
Explain the construction and content of a character table.
-
A student decides to give his bicycle a tune up. He flips it upside down (so there's no friction with the ground) and applies a force of 22 N over 1.6 seconds to the pedal, which has a length of 16.0...
-
= The charges and coordinates of two charged particles held fixed in an xy plane are q = 2.16 C, x = 4.49 cm, y = 0.925 cm and 92 -3.04 C, x2 -2.05 cm, y2 = 1.33 cm. Find the (a) magnitude and (b)...
-
Car A and car B travel in the same direction along a straight section of the interstate highway. For the entire interval shown on the velocity-versus-time graph (see figure below), car A is ahead of...

Study smarter with the SolutionInn App


