Consider the reaction TiO 2 (s) + 2 C(graphite) + 2 Cl 2 (g) 2 CO(g) +
Question:
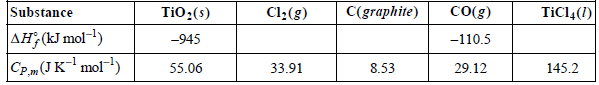
Assume that the heat capacities are independent of temperature.
a. Calculate ΔHoR at 135.8ºC, the boiling point of TiCl4.
b. Calculate ΔHof for TiCl4(l) at 25ºC.
Transcribed Image Text:
C(graphite) Co(g) TiCl4(1) TiO:(s) Cl:(g) Substance AH, (kJ mol-1) -110.5 -945 55.06 8.53 29.12 33.91 145.2 CP.(JKmol)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (7 reviews)
a In this case the heat capacities are assumed to be in...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
At 1000.K, H o R = 123.77 kJ mol 1 for the reaction N 2 (g) + 3H 2 (g) 2NH 3 (g), with C P,m = 3.502R, 3.466 R, and 4.217 R for N 2 (g), H 2 (g), and NH 3 (g), respectively. Calculate H o f of NH 3...
-
At 295 k, H o R = 131.28 kJ mol -1 for the reaction C (graphite) + H 2 O(g) CO(g), with C P,m = 8.53, 33.58, 29.12, and 28.82 J K -1 mol -1 for graphite. H 2 O(g), CO(g), and H 2 (g) respectively....
-
Calculate the standard enthalpy of formation of FeS 2 (s) at 600. °C from the following data at 298.15 K. Assume that the heat capacities are independent of temperature. You are also given that...
-
What is the timestep value ? And how do I go about altering from downstream to upstream? The following code solves the advection equation 1 2 3 4 5 6 7 8 9- 10 - 11 12 - 13 - 14 - 15 - 16 - 17 18 19...
-
The diagram in Figure 1 was developed for the project launch of Kitty Condo, a new product in the luxury cat cage market. Suppose that you, as project manager, are interested in finding ways to speed...
-
Daryl and Linda divorced two years ago, and Linda was given custody of their son, Thomas. During the current year, Daryl provided \($8,000\) of Thomass support, and Linda provided \($5,000. Which\)...
-
Carry out a full decision analysis for Classical Reproductions Ltd, using the following information: Calculation of expected profit with perfect information Prior probabilities for the various events...
-
Consider the following transactions that occurred in September 2012 for Aquamarines, Inc. Sep 3 Purchased inventory on terms 1/15, n/eom, $5,000. 4 Purchased inventory for cash of $1,700. 6 Returned...
-
We saw that if an economy has a negative shock to aggregate demand (maybe the COVID-19 lockdowns), that market forces will push the labor market back to equilibrium. Explain the steps that make this...
-
Alumni donations are an important source of revenue for colleges and universities. If administrators could determine the factors that influence increases in the percentage of alumni who make a...
-
Derive the equation (H/T) V = C V + V/ from basic equations and definitions.
-
Which of the following systems are open? a) A dog, b) An incandescent light bulb c) A tomato plant d) a can of tomatoes. Explain your answers.
-
What factors can cause overhead to be under-applied or over-applied? Are all of these factors controllable by management? Why or why not?
-
Undertake a critique of the different traditions of corporate governance in the UK/USA, Germany and Japan in terms of your own views of their strengths and weaknesses. Is there a better system than...
-
Wigit Construction has put in a bid to buy Wallocot Steel, one of its primary suppliers. What type of organizational change does this represent?
-
Gus is working in an oilfield assigned to a project in a remote area. The trailer he works out of has a telephone line, but Internet connectivity and mobile phone coverage is spotty. What would be...
-
What type of communication method would make sense for routine status meetings on a project where the team is spread out over different cities on the same continent?
-
In light of new tax incentives, a tech company incorporates in a different state and moves their headquarters. What type of organizational change does this represent?
-
The determinant of a 3 3 matrix A is defined as follows. The determinant of a 3 3 matrix can also be found using the method of diagonals. Step 1 Rewrite columns 1 and 2 of matrix A to the right of...
-
The Heese Restaurant Group manufactures the bags of frozen French fries used at its franchised restaurants. Last week, Heeses purchased and used 101,000 pounds of potatoes at a price of $ 0.70 per...
-
Calculate the speed to which a stationary 4He atom (mass 4.0026 u) would be accelerated if it absorbed each of the photons used in Exercise S.4b
-
A photon-powered spacecraft of mass 10.0 kg emits radiation of wavelength 225 nm with a power of 1.50 kW entirely in the backward direction. To what speed will it have accelerated after 10.0 y if...
-
A laser used to read CDs emits red light of wavelength 700 nm. How many photons does it emit each second if its power is? (a) 0.10 W, (b) LOW?
-
In the context of solid-liquid extraction, how do particle size distribution, porosity, and surface area of the solid matrix impact extraction kinetics and overall efficiency? Provide examples of...
-
Discuss the challenges and opportunities associated with the extraction of bioactive compounds from natural sources using environmentally benign solvents and green extraction techniques, such as...
-
The driver of a car traveling at 31.9 m/s applies the brakes and undergoes a constant deceleration of 1.12 m/s 2 . How many revolutions does each tire make before the car comes to a stop, assuming...

Study smarter with the SolutionInn App


