Consider two separate molar ensembles of particles characterized by the energy-level diagram provided in the text. Derive
Question:
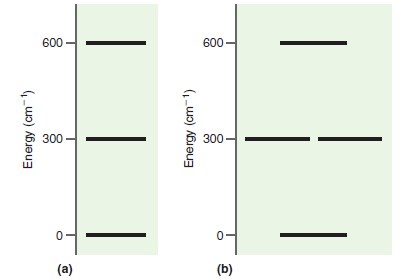
Transcribed Image Text:
600 600- 300 300- Energy (cm-1) Energy (cm-1)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
Evaluating the expressions for U A and U B a...View the full answer

Answered By

ROHIT JANGID
I just finished my M.Sc in Organic Chemistry. Though I didn't join any coaching center yet but I gave tuition at my own home. I used to start to teach student from basic of the topic by using examples of daily life as possible so that Science may become more easy to understand and interesting as well. I always try to focus on extending knowledge of any student, not only to solve any question or any problem only.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The energy level diagram for the MOs of CH 2 = CH 2 is shown if Figure 3.24. Show a similar diagram for the lowest-energy excited state of this molecule. C=C 5 o* * Ethene 5 o Energy
-
You are studying the kinetics of the reaction H2(g) + F2(g) 2HF(g) and you wish to determine a mechanism for the reaction. You run the reaction twice by keeping one reactant at a much higher pressure...
-
In a set of experiments on a hypothetical one-electron atom, you measure the wavelengths of the photons emitted from transitions ending in the ground state (n = I), as shown in the energy level...
-
A spring is at rest in the vertical direction. When a 5 kg mass is placed upon the spring, the length of the spring compresses to 0 . 2 0 meters. The 5 kg mass is removed and replaced by an 8 kg...
-
A page that is x inches wide and y inches high contains 30 square inches of print. The top and bottom margins are each 2 inches deep and the margins on each side are 2 inches wide. (a) Show that the...
-
A radial flow reactor is often used for highly exothermic reactions. The high radial velocities at the reactor inlet compensate for any hot spots that might form in the reactor there. Consider the...
-
What are the advantages and disadvantages of using the S-curve analysis as a project control tool?
-
Pal Corporation acquired 70 percent of the outstanding voting stock of Sal Corporation for $91,000 cash on January 1, 2011, when Sal's stockholders' equity was $130,000. All the assets and...
-
(a) Buyer contracts with Manufacturer for the production and delivery of 10,000 Texas Rangers jerseys which will be sold in Buyer's store as part of a special promotion. The contract price is...
-
Tuf-Stuff, Inc., sells a wide range of drums, bins, boxes, and other containers that are used in the chemical industry. One of the company's products is a heavy-duty corrosion-resistant metal drum,...
-
For a two-level system where v = 1.50 10 13 s 1 , determine the temperature at which the internal energy is equal to 0.25 Nh, or 1/2 the limiting value of 0.50 Nh.
-
For an ensemble consisting of a mole of particles having two energy levels separated by 1000. cm 1 , at what temperature will the internal energy equal 3.00 kJ?
-
The data shown are the weekly admissions, in millions, of people attending movie theaters over three different time periods. At α = 0.05, is there a difference in the means for the...
-
Braverman Company has two manufacturing departments-Finishing and Fabrication. The predetermined overhead rates in Finishing and Fabrication are $28.00 per direct labor-hour and 120% of direct...
-
A company is faced with the decision of whether to lease or purchase new equipment. The equipment can be leased at an annual cost of $210,000 or purchased for $560,000. The equipment has an expected...
-
John wants to "roll in" or finance the loan fee of $3,800 into the loan amount which would make the loan $90,800 and the interest rate is 6%. Assume that the lender agrees to allow the loan fees to...
-
Johnny's Lunches is considering purchasing a new, energy-efficient grill. The grill will cost $31,000 and will be depreciated straight-line over 10 years to a salvage value of zero. The grill will...
-
Inflation in the United States is 2% per year and in Australia, it is 3% per year. The nominal interest rate in Australia is 6%. What does the Fisher effect predict the nominal interest rate in the...
-
When gaining an understanding of the client at the industry level, the auditor: (a) Will not ignore information about the clients industry. (b) Will not consider the level of demand for the goods and...
-
Nike manufactures shoes and sportswear. How has the Internet changed the way this company communicates with its suppliers and retail customers?
-
The following compound is optically inactive. Explain why.
-
Identify the reagent you would use to accomplish each of the following transformations: (a) Cyclobutanol bromocyclobutane (b) tert-Butanol tert-butyl chloride (c) Ethyl chloride ethanol
-
How many different alkenes will produce 2, 4-dimethylpentane upon hydrogenation? Draw them.
-
A child jumps from a moving sled with a speed of 2.2 m/s and in the direction opposite the sleds motion. The sled continues to move in the forward direction, but with a new speed of 5.5 m/s. If the...
-
An iron block with a mass of 45 000 g rests on an incline plane of 16 to the horizontal. Take the coefficient friction as 0,25. Calculate the following: 1.1 the weight component perpendicular to the...
-
29. You are standing on a balcony on the 15th floor of the Bank of America building in San Francisco. The balcony is 72 m above the ground; the top of the building is 52 floors (237 m) high. You...

Study smarter with the SolutionInn App


