Determine the half-cell reactions and the overall cell reaction, calculate the cell potential, and determine the equilibrium
Question:
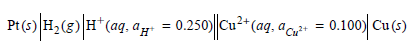
Is the cell reaction spontaneous as written?
Transcribed Image Text:
PrO|#,(e)|#"(aq, ayr = 0.250||0a*(aq. aar• = 0.100| Cu() - 0.100) Cu() = 0.250) Cu²* (ag, a c: = 0.100) Cu (s) Pt(:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (6 reviews)
We first calculate the E o for the reaction Cu 2 aq 2e Cus from the ...View the full answer

Answered By

Akshay Agarwal
I am a Post-Graduate with a specialization in Finance. I have been working in the Consulting industry for the past 8 years with a focus on the Corporate and Investment Banking domain. Additionally, I have been involved in supporting student across the globe in their academic assignments and always strive to provide high quality support in a timely manner. My notable achievements in the academic field includes serving more than 10,000 clients across geographies on various courses including Accountancy, Finance, Management among other subjects. I always strive to serve my clients in the best possible way ensuring high quality and well explained solutions, which ensures high grades for the students along-with ensuring complete understanding of the subject matter for them. Further, I also believe in making myself available to the students for any follow-ups and ensures complete support and cooperation throughout the project cycle. My passion in the academic field coupled with my educational qualification and industry experience has proved to be instrumental in my success and has helped me stand out of the rest. Looking forward to have a fruitful experience and a cordial working relationship.
5.00+
179+ Reviews
294+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
At 298 K a cell reaction has a standard cell potential of +0.17 V. The equilibrium constant for the reaction is 5.5 105. What is the value of n for the reaction?
-
The overall reaction and standard cell potential at 25oC for the rechargeable nickelcadmium alkaline battery is Cd(s) + NiO2(s) + 2H2O(l) Ni(OH)2(s) + Cd(OH)2(s) o = 1.10 V For every mole of Cd...
-
Determine the equilibrium constant for the reaction CH4 + 2O2 CO2 + 2H2O when the reaction occurs at 100 kPa and 2000 K. The natural logarithms of the equilibrium constant for the reaction C + 2H2...
-
Harvold Company's quality cost report is to be based on the following data: Test and inspection of incoming materials. $71,000 Supplies used in testing and inspection . Re-entering data because of...
-
Wheatmore Company manufactures cold cereal products. Assume that the inventory in process on January 1 for the Packing Department included 1,290 pounds of cereal in the packing machine hopper, enough...
-
Police officers arrived at Grants home to investigate a tip that Grants residence was being used to sell drugs. At the time, Grant was not at home, but he did arrive home in his car while police were...
-
For a gas turbine the pressure ratio may be in the range (a) 2 to 3 (b) 3 to 5 (c) 6 to 10 (d) 12 to 16
-
The CPA firm of Quigley & Associates uses a qualitative approach to implementing the audit risk model. Audit risk is categorized using two terms: very low and low. The risk of material misstatement...
-
Blood flows through the aorta at an average speed of v = 19cm/s. The aorta is roughly cylindrical with a radius r = 17.6 mm. The volume rate of blood flow through the aorta is r 2 v . Calculate the...
-
Figure P2.26 a shows a uniform beam subject to a linearly increasing distributed load. As depicted in Fig. P2.26 b , deflection y (m) can be computed with where E = the modulus of elasticity and I =...
-
Table 2.10 presents data on mean SAT reasoning test scores classified by income for three kinds of tests: critical reading, mathematics, and writing. In Example 2.2, we presented Figure 2.7, which...
-
What is the conditional expectation function or the population regression function?
-
Find the area between a large loop and the enclosed small loop of the curve r = 1 + 2 cos 3.
-
Describe how the legislative branch of government is a source of law.
-
Which of the following best describes situations with complex frauds and financial crimes? 1. Organizations associated with complex frauds and financial crimes often dismantle legitimate businesses....
-
The Investment Company Act of 1940 prohibits a mutual fund from engaging in certain transactions in which there may be a conflict of interest between the manager of the fund and its shareholders....
-
An engineer had formerly been involved with manufacturing nuclear reactor parts that were to be put onto U.S. Navy vessels. However, he was fired after superiors heard he had been complaining to...
-
What are the defendants Miranda rights?
-
Which number is closest to Avogadros number? (a) The number of people on the Earth. (b) The number of stars in our galaxy. (c) The number of O 2 molecules in your lungs. (d) The number of hairs on...
-
With your classmates, form small teams of skunkworks. Your task is to identify an innovation that you think would benefit your school, college, or university, and to outline an action plan for...
-
The hyperfine coupling constants observed in the radical anions (12), (13), and (14) are shown (in millitesla, mT). Use the value for the benzene radical anion to map the probability of finding the...
-
The chemical shift of the CH 3 protons in acetaldehyde (ethanal) is = 2.20 and that of the CHO proton is 9.80. What is the difference in local magnetic field between the two regions of the molecule...
-
The first generally available NMR spectrometers operated at a frequency of 60 MHz; today it is not uncommon to use a spectrometer that operates at 800 MHz. What are the relative population...
-
Direct materials Fixed manufacturing overhead costs Sales price Variable manufacturing overhead Direct labor Fixed marketing and administrative costs Units produced and sold during the quarter...
-
Provide documentation of a step by step training plan for a beta fish. What will be the primary reinforcer? How will you cue the behavior? What schedule of reminforcement will you use. What is the...
-
On May 1, Soriano Company reported the following account balances along with their estimated fair values: Items Carrying Amount Fair Value Receivables Inventory Copyrights Patented technology Total...

Study smarter with the SolutionInn App


