In an experiment on the Pt|H 2 |H + electrode in dilute H 2 SO 4 the
Question:
In an experiment on the Pt|H2|H+ electrode in dilute H2SO4 the following current densities were observed at 25°C. Evaluate α and j0 for the electrode.
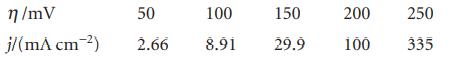
How would the current density at this electrode depend on the overpotential of the same set of magnitudes but of opposite sign?
Transcribed Image Text:
η/mV j/(m^ cm-²) 50 100 150 2.66 8.91 29.9 200 100 250 335
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 16% (6 reviews)
Solution Using the equations for PtH2H and PtH2H in dilute H2SO4 we have Now b...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
In an experiment on the Pt IH,I H+ electrode in dilute H,S04 the following current densities were observed at 25e. Evaluate ET and jo for the electrode. How would the current density at this...
-
The following data were observed in an experiment on the photoelectric effect from potassium: Graphically evaluate these data to obtain values for the work function and Plancks constant. 1019 Kinetic...
-
In an experiment on soybean varieties, individually potted soybean plants were grown in a greenhouse, with 10 plants of each variety used in the experiment. From the harvest of each plant, five seeds...
-
Differentiate implicitly to find 2x + 5xy + 5y +16y - 8 = 0; dy dx . Then find the slope of the curve at the given point. (-2,0)
-
The data in the file named Fast100 were collected by D. L. Green & Associates, a regional investment management company that specializes in working with clients who wish to invest in smaller...
-
Two forces F1 and F2 act on the bolt. If the resultant force FR has magnitude FR and coordinate direction angles α and β, as shown, determine the magnitude of F2 and its coordinate...
-
What are the main similarities and differences between BS 7750, EMAS and ISO 14001? Is one of these systems to be preferred over the other two?
-
An ammonia solution at a high pressure is flash-vaporized at a rate of 200lbm/h. The solution contains 0.70lbm NH3/lbm, and its enthalpy relative to H2O (1, 32F) and NH3 (1, 40F) is 50 Btu/lbm....
-
The borrower will have received monthly payments of $1,382.50 during months 1 to 36 Solve for loan balance at the end of month 36
-
Table 14.1 was constructed under the assumption that all firms in the industry are identical. A: Suppose that all firms in an industry have U-shaped long run average cost curves. (a) Leaving aside...
-
In an experiment on the adsorption of oxygen on tungsten it was found that the same volume of oxygen was desorbed in 27 min at 1856 K and 2.0 min at 1978 K. What is the activation energy of...
-
Nitrogen gas adsorbed on charcoal to the extent of 0.921 cm 3 g 1 at 490 kPa and 190 K, but at 250 K the same amount of adsorption was achieved only when the pressure was increased to 3.2 MPa. What...
-
Why is it important to consider the relationships among cost, quality, and selling prices when establishing standards for direct materials?
-
You would like to know what the market expects the 1 year rate will be three years from now. You observe the following prices for pure discount, or zero-coupon, bonds: Maturity Zero Coupon Bond Price...
-
If w=.108% find dia J2 and determine whene wis non-analytic.
-
The solution of ly - 15| <23 is
-
Propose some storage system alternatives that you could consider for long term storage of geotechnical design documentation.?
-
The "Do not sell fireplaces in Hawaii." Company expects their dividends and earnings to grow at a constant rate of 3% a year into the foreseeable future. Currently the market is requiring a 7.3% rate...
-
The box on page 314 describes Singapore Airlines option to buy additional A350 airliners. Draw a decision tree showing the future choices faced by the airline.
-
A business had revenues of $280,000 and operating expenses of $315,000. Did the business (a) Incur a net loss (b) Realize net income?
-
One mole of H 2 O(l) is super cooled to 3.75C at 1 bar pressure. The freezing temperature of water at this pressure is 0.00C. The transformation H 2 O(l) H 2 O(s) is suddenly observed to occur. By...
-
An athlete at high performance inhales ~3.75 L of air at 1.0 atm and 298 K at a respiration rate of 32 breaths per minute. If the exhaled and inhaled air contain 15.3 and 20.9% by volume of oxygen,...
-
The temperature of 1.75 moles of an ideal gas increases from 10.2C to 48.6C as the gas is compressed adiabatically. Calculate q, w, U, and H for this process assuming that C V ,m = 3/2 R.
-
In eukaryotes, which two processes occur during termination of transcription?
-
Below is the list of eukaryotic organelles. Mitochondria | Plant chloroplast | Nucleus | Lysosome | Nuclear envelope | Rough endoplasmic reticulum | Golgi apparatus | Smooth endoplasmic reticulum |...
-
Study customer touchpoints. Remember that we are working with the B2C market. Select two stages of the customer decision journey Answer the following questions: For each selected stage, identify a...

Study smarter with the SolutionInn App


