From the following data at 298.15 K calculate the standard enthalpy of formation of FeO(s) and of
Question:
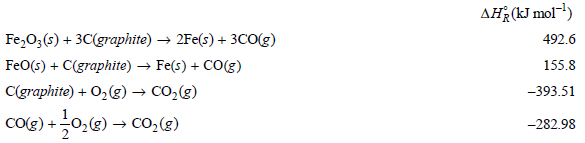
Transcribed Image Text:
AĦ¿(kJ mol) Fe,0;(s) + 3C(graphite) → 2Fe(s) + 3cO(g) FeO(s) + C(graphite) → Fe(s) + CO(g) C(graphite) + O2(g) → CO,(g) CO(e) +0,@) → Co,(3) 492.6 155.8 -393.51 -282.98
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (15 reviews)
Fes COg FeOs Cgraphite COg 1 0 8 Cgraphite Og COg ...View the full answer

Answered By

Gauri Hendre
I worked as EI educator for Eduphy India YT channel. I gave online tutorials to the students who were living in the villages and wanted to study much more and were preparing for NEET, TET. I gave tutions for topics in Biotechnology. I am currently working as a tutor on course hero for the biochemistry, microbiology, biology, cell biology, genetics subjects. I worked as a project intern in BAIF where did analysis on diseases mainly genetic disorders in the bovine. I worked as a trainee in serum institute of India and Vasantdada sugar institute. I am working as a writer on Quora partner program from 2019. I writing on the topics on social health issues including current COVID-19 pandemic, different concepts in science discipline. I learned foreign languages such as german and french upto A1 level. I attended different conferences in the science discipline and did trainings in cognitive skills and personality development skills from Lila Poonawalla foundation. I have been the member of Lila poonawalla foundation since 2017. Even I acquired the skills like Excel spreadsheet, MS Office, MS Powerpoint and Data entry.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Calculate the standard enthalpy of formation for diamond, given that C(graphite) + O2(g) CO2(g) Afr =-393.5 kJ/mol C(diamond) + O2(g) CO2(g) AF1 395.4 kJ/mol
-
From the following data at 298.15 K as well as data in Table 4.1 (Appendix B, Data Tables), calculate the standard enthalpy of formation of H 2 S(g) and of FeS 2 (s): AR(kJ mol) Fe(s) + 2H2S(g) ...
-
Calculate the standard enthalpy of formation of FeS 2 (s) at 600. °C from the following data at 298.15 K. Assume that the heat capacities are independent of temperature. You are also given that...
-
Suppose that today, you paid $1,000 for the bond described in Problem 8 of Chapter 8, The net present value functions: NPV and XNPV. What would be the bonds IRR? A bonds IRR is often called the yield...
-
Formulating an Argument about Earlier American Literature "What question at issue concerning earlier American literature has your research enabled you to identify and answer?" Looking ahead to the...
-
Under what conditions is a process-costing system used?
-
Explain the meaning of the following terms in variables sampling: (a) precision, (b) reliability, and (c) variability.
-
Moreno Industries has adopted the following production budget for the first 4 months of 2011. Each unit requires 3 pounds of raw materials costing $2 per pound. On December 31, 2010, the ending raw...
-
Claudia is self-employed and earns $190,000 p.a. If she makes a tax deductible (concessional) contribution to superannuation of $15,000, will she save tax and if so, how much? Exclude Medicare Levy/...
-
Which of the following statements are false? 1. Revenue provides only outward flows of cash. 2. Revenue is a subdivision of Assets. 3. Revenue provides an inward flow of cash or accounts receivable....
-
Compare the heat evolved at constant pressure per mole of oxygen in the combustion of sucrose (C 12 H 22 O 11 ) and palmitic acid (C 16 H 32 O 2 ) with the combustion of a typical protein, for which...
-
A camper stranded in snowy weather loses heat by wind convection. The camper is packing emergency rations consisting of 58% sucrose, 31% fat, and 11% protein by weight. Using the data provided in...
-
Develop a two-period weighted moving average forecast for March 2016 through January 2017. Use weights of 0.6 and 0.4, with the most recent observation weighted higher. Calculate the MFE, MAD, and...
-
Computing and Comparing Income and Cash Flow MeasuresPenno Corporation recorded service revenues of $ 1 2 0 , 0 0 0 in 2 0 2 0 , of which $ 1 0 2 , 0 0 0 were on credit and $ 1 8 , 0 0 0 were for...
-
Jasper and Crewella Dahvill were married in year 0 . They filed joint tax returns in years 1 and 2 . In year 3 , their relationship was strained and Jasper insisted on filing a separate tax return....
-
A corporation is working on its cash budget for March. The budgeted beginning cash balance is $ 3 3 , 0 0 0 . Budgeted cash receipts total $ 1 8 2 . 0 0 0 and budgeted cash disbursements total $ 1 9...
-
Bonds payable record issuance and premium amortization Jessie Co . issued $ 3 million face amount of 9 % , 2 0 - year bonds on July 1 , 2 0 2 2 . The bonds pay interest on an annual basis on June 3 0...
-
Simpson, age 4 5 , is a single individual who is employed full time by Duff Corporation. This year Simpson reports AGI of $ 5 5 , 2 0 0 and has incurred the following medical expenses: Dentist...
-
Give five other iterated integrals that are equal to 22 a, y, z) dz dx dy 10 J0
-
Which of the companies has the lowest accounts receivable turnover in the year 20X2? a. Company A. b. Company B. c. Company C. d. CompanyD. 20X1 20X2 Credit Sales Average Receivables Balance $1.0...
-
For how long on average would an atom remain on a surface at 400 K if its desertion activation energy were? (a) 20 kO mol-1, (b) 200 k] mol-I? Take TO = 0.12 ps. For how long on average would the...
-
A solid in contact with a gas at 8.86 kPa and 25C adsorbs 4.67 mg of the gas and obeys the Langmuir isotherm. The enthalpy change when 1.00 mmol of the adsorbed gas is desorbed is +12.2 J. What is...
-
Suppose it is known that ozone adsorbs on a particular surface in accord with a Langmuir isotherm. How could you use the pressure dependence of the fractional coverage to distinguish between...
-
The following are extracted from the Cash Book of Jungle Trading and its Bank Statement (received from ABC Bank) for the month of April 2020: Cash Book April 2020 RM April 2020 1 Bal b/d 1,500 9...
-
1. Briefly describe FIVE (5) differences between qualitative data and quantitative data. 2. The literature review is an integral part of the research process and make a valuable contribution to...
-
Writing a detailed written assessment that challenges the traditional legal regimes governing the use of military force, addressing key jus ad bellum and jus in bello legal concerns along with their...

Study smarter with the SolutionInn App


