Predict whether the ground state or the first excited state of CH 2 should have the larger
Question:
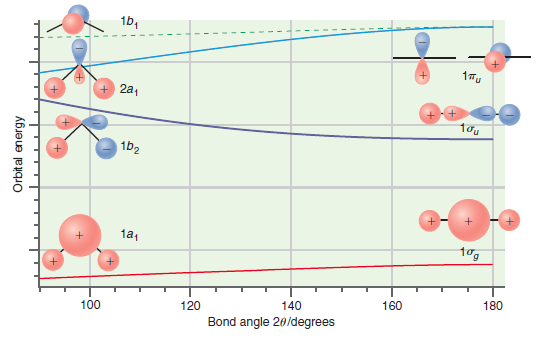
Transcribed Image Text:
1b, 17u 2a, 1b2 1a, tog 180 160 140 120 100 Bond angle 20/degrees Orbit al energy
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
CH 2 has six valence electrons In the ground state the HOMO is the 2a ...View the full answer

Answered By

Ankit Mahajan
I am an electrical engineering graduate from Thapar institute of engineering and technology.
Qualified exams - GATE 2019,2020.
CAT EXAM 2021- 91.4 percentile
SSC EXAMS- 2019,2020,2021
AFCAT EXAM- 2019,2020,2021
I want to share my knowledge with other people so that they can achieve the same.
I have strong hold Mathematics, Electrical engineering and all the subjects related.
Just give me a problem and I will give you the solution of it.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict which of the bent molecules, BH 2 or NH 2 , should have the larger bond angle on the basis of the Walsh correlation diagram in Figure 24.11. Explain your answer. Figure 24.11 16, 1Tu + 2a,...
-
Explain whether the electron arrangement for these atoms is the ground state or an excitedstate: a) Energy 3 Nitrogen b) Energy 2p 5 4 Carbon
-
Predict whether LiH + 2 and NH 2 should be linear or bent based on the Walsh correlation diagram in Figure 24.11. Explain your answers. Figure 24.11 1b, + 2a, 1b2 1a1 tog 100 120 140 160 180 Bond...
-
In an organization that has high employee satisfaction, ______. Multiple choice question. customer interactions are forced and scarce employee turnover is high more positive interactions take place...
-
Consider an angle in standard position with r = 12 centimeters, as shown in the figure. Describe he changes in the values of x, y, sin θ, cos θ, and tan θ as...
-
List and briefly explain the different purchase options available to anyone who wants to consider buying a business.
-
What are COTS products and NDIs and how do they relate to items and CIs?
-
Ursula Company maintains a petty cash fund for small expenditures. The following transactions occurred over a 2-month period. July 1 Established petty cash fund by writing a check on Scranton Bank...
-
Explain why the circuit in Figure 4 is a 4-to-1 MUX. 85 W W Vo Yo V1 y 12 W2 1 En D D Figure 4 f
-
In addition to the information for Acme Company in Mini-Exercises 15.1 and 15.2, the standard direct labor cost per unit consists of 0.5 hour of labor time at $20 per hour. During August, $160,650 of...
-
The occupied MOs of ammonia are shown next along with the MO energies. Indicate which AOs are most important in each MO and indicate the relative phases of the AOs. Classify the MOs as localized or...
-
The occupied MOs of ethene are shown next along with the MO energies. Indicate which AOs are most important in each MO and indicate the relative phases of the AOs. Classify the MOs as localized or...
-
What are the (a) Frequency, in s -1 , and (b) Wavelength, in nanometers, of the light emitted when the electron in a hydrogen atom drops from the energy level n = 7 to n = 4? (c) In what portion of...
-
2. The stockholders' equity of A&C Company consisted of the following balances: Common Stock, $10 par, authorized 300,000 shares issued and outstanding 248,000 shares APIC - Common Preferred 5%...
-
Creative Technology reports inventory using the lower of cost and net realizable value (NRV). Below is information related to its end inventory. year- Inventory Optima cameras Unit Unit Quantity Cost...
-
The Jurassic Classics has four employees on its sales team and uses a compensation that provides each person with a base salary of $40,800 per year and the opportunity to earn commission on sales....
-
How many segments are there from which to target? Which segment should Evan target? How to find VIU for the commercial user segment because of its VIU score .-Industrial users VIU and Residential...
-
E3-16 The December 31, 2024, adjusted trial balance for Fightin Blue Hens Corporation is presented below. Required: Accounts Debit Credit Cash $ 12.000 Accounts Receivable 150,000 Prepaid Rent 6,000...
-
Spartan Builders Corporation is a builder of high-end housing with locations in major metropolitan areas throughout the Midwest. At June 30, 2018, the company has deferred tax assets totaling $10...
-
Ex. (17): the vector field F = x i-zj + yz k is defined over the volume of the cuboid given by 0x a,0 y b, 0zc, enclosing the surface S. Evaluate the surface integral ff, F. ds?
-
A second-order reaction of the type A + B P was carried out in a solution that was initially 0.050 mol dm 3 in A and 0.080 mol dm 3 in B. After 1.0 h the concentration of A had fallen to 0.020 mol...
-
Cyclopropane isomerizes into propene when heated to 500C in the gas phase. The extent of conversion for various initial pressures has been followed by gas chromatography by allowing the reaction to...
-
The rate constant for the first-order decomposition of N 2 O 5 in the reaction 2 N 2 O 5 (g) 4 NO 2 (g) + O 2 (g) is k = 3.38 10 5 s 1 at 25C. What is the half-life of N 2 O 5 ? What will be the...
-
A 0.55 kg block of ice is sliding by you on a very slippery floor at 3.5 m/s. As it goes by, you give it a kick perpendicular to its path. Your foot is in contact with the ice block for 0.0035...
-
Write a script called ChangeExtensions that accepts two file name extensions as arguments, and renames all files with the first extension to have the second extension. For example, Change Extensions...
-
Write an HTML document that draws the trapezoid (a rectangle that is wider on one side) below in Canvas. (30,30) 50 50 70

Study smarter with the SolutionInn App


