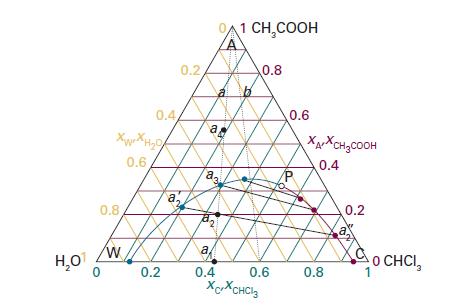
Refer to the ternary phase diagram in Fig. 5D.4. How many phases are present, and what are
Question:
Refer to the ternary phase diagram in Fig. 5D.4. How many phases are present, and what are their compositions and relative abundances, in a mixture that contains 2.3 g of water, 9.2 g of chloroform, and 3.1 g of acetic acid? Describe what happens when (i) water, (iii) acetic acid is added to the mixture.
Data in Fig. 5D.4.
Transcribed Image Text:
H₂O Xuan Ho 0.6 0.8 0.4 W 0.2 a 20 0.2 a a. 23 a 0.4 1 CH₂COOH 0.8 0.6 XCXCHCl3 0.6 XAXCH₂COOH 0.4 0.8 0.2 O CHCI
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
For the given mixture there are three phases present wat...View the full answer

Answered By

BillClinton Muguai
I have been a tutor for the past 5 years. I have experience working with students in a variety of subject areas, including computer science, math, science, English, and history. I have also worked with students of all ages, from elementary school to college. In addition to my tutoring experience, I have a degree in education from a top university. This has given me a strong foundation in child development and learning theories, which I use to inform my tutoring practices.
I am patient and adaptable, and I work to create a positive and supportive learning environment for my students. I believe that all students have the ability to succeed, and it is my job to help them find and develop their strengths. I am confident in my ability to tutor students and help them achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Physical Chemistry Thermodynamics And Kinetics
ISBN: 9781464124518
10th Edition
Authors: Peter Atkins, Julio De Paula
Question Posted:
Students also viewed these Sciences questions
-
A mixture that contains 46 wt% acetone (CH3COCH3), 27% acetic acid (CH3COOH), and 27% acetic anhydride [(CH3CO) 2O] is distilled at P = 1 atm. The feed enters the distillation column at T = 348 K at...
-
How many phases are present at each of the points marked in Fig. 4.1a? Data in Fig. 4.1a? Pressure (a) d b Temperature O a
-
A mixture that contains benzoic acid, 4-chloroaniline and naphthalene. The 4-chloroaniline is separated first by extraction with hydrochloric acid.Since no phenolic compound is present in this...
-
Consider the following velocity distribution curves A and B. a. If the plots represent the velocity distribution of 1.0 L of He(g) at STP versus 1.0 L of Cl2(g) at STP, which plot corresponds to each...
-
Does recognizing an impairment loss on a long- term operating asset have the same effect on the financial statements as recording depreciation expense and amortization expense? Explain.
-
Flanagan Company reports the following for the month of June. Instructions(a) Calculate the cost of the ending inventory and the cost of goods sold for each cost flow assumption, using a perpetual...
-
The following MINITAB output presents a multiple regression equation y = b0 + b1x1 + b2x2 + b3x3. Test H0 : i = 0 versus H1: i 0 for i = 1, 2, 3. Use the = 0.05 level. The regression equation is...
-
Assume a Gold Medal Sports outlet store began August 2012 with 44 pairs of running shoes that cost the store $33 each. The sale price of these shoes was $61. During August, the store completed these...
-
Please complete the answers in the charts format? The following information pertains to Flaxman Manufacturing Company for April. Assume actual overhead equaled applied overhead. April 1 Inventory...
-
From the data 1,4, 5, 3, 2, 5, 7, 3, 4, and 5, Poindexter created the following frequency table. What five things did he do wrong? f- 013589 f- 11223|-| 123457
-
Use mathematical software, a spreadsheet, or the Living graphs on the web site for this book to plot p A /p A * against x A with =2.5 by using eqn 5E.19 and then eqn 5E.20. Above what value of x A...
-
Could a regular tetrahedron be used to depict the properties of a four component system?
-
What is customer-centric new product development? Provide an example.
-
Given f (4) = 2, f' (4) 10, g (4) = -1, and g' (4) 7, find the values of the following. 9 (b) HIST (fg)' (4) = Number 4) (4)- Lensessan 9 Number
-
Given the following situation of a marble undergoing constant velocity on a rail: Down: v = 10. m/s. h = 1.5 m . A) Sketch a motion map showing the motion of the marble after it leaves the rail. Show...
-
B ased budgeting focuses on how products are produced rather than what inputs are purchased and how much output is produced?
-
8% Intermediate Accounting Co. Shareholders' equity 31-Dec-19 Preferred shares, no par value, cumulative and participating 1,000 shares issued & outstanding Common shares, no par value, 100,000...
-
Preparing a Classified Balance Sheet The following financial data for Crane Distributors was collected as of December 3 1 . Cash $ 1 5 , 2 0 0 Delivery equipment $ 8 0 , 0 0 0 Accounts receivable 1...
-
Recompute the Gram matrices for cases (vi)-(viii) in Exercise 3.4.22 for the weighted inner product (x,y) = x1,y1, + 1/2x2y2 + 1/3X3y3 + 1/4 x4y4.
-
Refer to the situation described inBE 18-13, but assume a 2-for-1 stock split instead of the 5% stock dividend. Prepare the journal entry to record the stock split if it is to be effected in the form...
-
Use the relation the cyclic rule and the van der Waals equation of state, to derive an equation for C P,m C V,m in terms of V m , T, and the gas constants R, a, and b. aV . Cr m = T
-
The amino acid glycine dimerizes to form the dipeptide glycylglycine according to the reaction 2Glycine(s): Glycylglycine(s) + H 2 O(l) Calculate ÎS, ÎS surr , and ÎS suniverse at T...
-
A 3.75 mole sample of an ideal gas with C V ,m = 3/2 R initially at a temperature T i = 298 K and P i = 1.00 bar is enclosed in an adiabatic piston and cylinder assembly. The gas is compressed by...
-
Needs Analysis : The analysis of the information gathered on the community military veterans and mental health. This will provide a conclusions you have drawn from thinking about the findings you...
-
How do I calculate the Portfolio Expected Return: You own aportfolio that has $4,600 invested in Stock X and $5,200 investedin Stock Z. What is the expected return on the portfolio if theexpected...
-
Students are asked to assume that during an advanced degree program, they thoroughly studied and reviewed the most popular personality tests used by companies as selection techniques. Students are...

Study smarter with the SolutionInn App


