Show that the determinantal property used in the discussion of localized and delocalized orbitals in Section 24.6
Question:
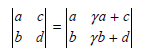
used in the discussion of localized and delocalized orbitals in Section 24.6 is correct.
Transcribed Image Text:
ya + c a ь la b yb + d 9.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
We first assume that for some con...View the full answer

Answered By

Pushpinder Singh
Currently, I am PhD scholar with Indian Statistical problem, working in applied statistics and real life data problems. I have done several projects in Statistics especially Time Series data analysis, Regression Techniques.
I am Master in Statistics from Indian Institute of Technology, Kanpur.
I have been teaching students for various University entrance exams and passing grades in Graduation and Post-Graduation.I have expertise in solving problems in Statistics for more than 2 years now.I am a subject expert in Statistics with Assignmentpedia.com.
4.40+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Why cant localized orbitals be represented in an MO energy diagram?
-
Why are localized and delocalized models equally valid for describing bonding in closed-shell molecules? Why cant experiments distinguish between these models?
-
The occupied MOs of ammonia are shown next along with the MO energies. Indicate which AOs are most important in each MO and indicate the relative phases of the AOs. Classify the MOs as localized or...
-
Terminology Key: Key Word List Describe Explain What to do In bulleted, numbered or paragraph form, provide a number of consecutive items-if paragraph form, use commas (,) to separate items In...
-
Find two solutions of each equation. Give your answers in degrees (0o < 360o) and in radians (0 < 2). Do not use a calculator. 1. (a) sin = 1/2 (b) sin = 1/2 2. (a) cos = 2/2 (b) cos = 2/2
-
What are the partners' rights and responsibilities?
-
What is a NDI?
-
Pace Company owns 85% of the outstanding common stock of Sand Company and all the outstanding common stock of Star Company. During 2012, the affiliates engaged in intercompany sales as follows: Sales...
-
Explain how public unrest, power outages, and severe weather could impact the metropolitan capital of Iowa (Des Moines) in regards to emergency management. 2. Explain what are the goals of an...
-
There is a lottery with n coupons and n people take part in it. Each person picks exactly one coupon. Coupons are numbered consecutively from 1 to n, n being the maximum ticket number. The winner of...
-
A cyclic polyene is known to be non-planar. Are the MO energy levels of this molecule well described by the Huckel model? Justify your answer.
-
Predict whether LiH + 2 and NH 2 should be linear or bent based on the Walsh correlation diagram in Figure 24.11. Explain your answers. Figure 24.11 1b, + 2a, 1b2 1a1 tog 100 120 140 160 180 Bond...
-
Various contractual arrangements entered into by Nusbaum Company require it to maintain a minimum balance of cash and cash equivalents of \($1\),000,000. The company's balance is dipping near that...
-
A population of animals has a per-capita birth rate of b = 0.17 per year and a per-capita death rate of m 0.1 per year. The population density. P(t) is found to satisfy the differential equation...
-
When a 20.0-V battery is connected to a capacitor, it stores a charge of 650C. What is the value of the capacitance (unit in F)?
-
C Four capacitors are connected to a DC battery (with an electric potential difference of V) as shown in Figure. Given: HH C2 C3 C1 (F) C2 (F) C3 (FC4 (F) V (Volt) C4 24 61 79 32 14 Calculate the...
-
A nucleus of the boron11 isotope consists of five protons and six neutrons. A particular ionized atom of boron11, whose mass is 1.831026kg,1.831026kg, lacks 33 electrons from its neutral state. Find...
-
Sheridan Company owns 41480 of the 103700 outstanding shares of Splish Inc. common stock. During 2022, Splish earns $663680 and pays cash dividends of $497760. If the beginning balance in Sheridan's...
-
Explain when an acquiring corporation would prefer to buy the target corporations assets directly in an acquisition.
-
The Ranch 888 Noodle Company sells two types of dried noodles:ramen, at $6.50 per box, and chow fun, at $7.70 per box. So farthis year, the company has sold a total of 110,096 boxes ofnoodles,...
-
What are the relative populations of the states of a two-level system when the temperature is infinite?
-
The element polonium crystallizes in a cubic system. Bragg reflections, with X-rays of wavelength 154 pm, occur at sin = 0.225, 0.316, and 0.388 from the (100), (110), and (111) sets of planes. The...
-
A 1.0 dm 3 glass bulb contains 1.0 10 2 3 H 2 molecules. If the pressure exerted by the gas is 100 kPa, what are (a) The temperature of the gas, (b) The root mean square speeds of the molecules? (c)...
-
of estion 2.1 Write a C++ program that calculates a customer's bill for a local cable company. The cable company has two types of customers: residential and business with different rates allocated...
-
Use Sobel mask and find horizontal and vertical gradient outputs for the given 5x5 image matrix which is coded 4-bit/pixel. (Don't use padding operation, obtain your output matrix as 3x3 image size!)...
-
003 The minimal polynomial associated with the matrix 102 01 1 is

Study smarter with the SolutionInn App


