Predict whether LiH + 2 and NH 2 should be linear or bent based on the Walsh
Question:
Figure 24.11
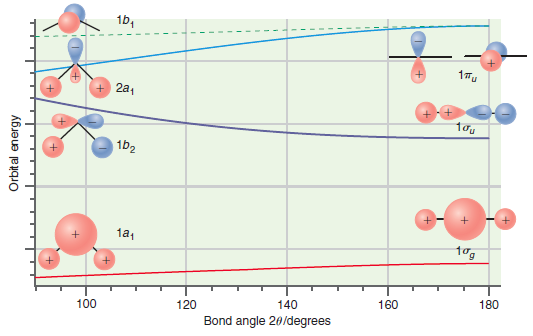
Transcribed Image Text:
1b, + 2a, 1b2 1a1 tog 100 120 140 160 180 Bond angle 20/degrees Orbital energy
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (13 reviews)
The LiH 2 molecular ion has two valence electrons Th...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict whether the ground state or the first excited state of CH 2 should have the larger bond angle on the basis of the Walsh correlation diagram shown in Figure 24.11. Explain your answer. 1b, 17u...
-
BeH 2 is linear, whereas CH 2 with two additional electrons and H 2 O with four additional electrons are both bent to a similar degree. Could these changes in geometry have been anticipated by...
-
Based on the table, if the temperature changes from 25°C to 65°C in a 105-m section of 9.5 Ã 103-m diameter wire, approximately how much will the wire's resistance change? A. 0.04...
-
Part 2: Use the activity-based information below to calculate the cost of each unit of product using machine setups instead of machine hours. Cost Driver Number of Machine Setups Operating Machines...
-
Use a calculator to evaluate the trigonometric function. Round your answer to four decimal places. (Be sure the calculator is in the correct mode.) 1. sin 10 2. tan 304 3. cos(110) 4. sin(330) 5. cot...
-
Develop a formula to compute the RMS bandwidth of a random process, X (t), directly from its autocorrelation function, RXX ().
-
The Principles for Responsible Investment (PRI) in their ESG integration in listed equity: A technical guide (2023) defines ESG integration as the process of including ESG factors in investment...
-
Prepare a report for the dean of the college that summarizes your assessment of the nature of cheating by business students at Bayview University. Be sure to include the following items in your...
-
Smith Electronic Company's chip-mounting production department had 300 units of unfinished product, each 50% completed on September 30. During October of the same year, this department put another...
-
Sunbright Citrus Products produces orange juice, grapefruit juice, and other citrus-related items. Sunbright obtains fruit concentrate from a cooperative in Orlando consisting of approximately 50...
-
Show that the determinantal property used in the discussion of localized and delocalized orbitals in Section 24.6 is correct. ya + c a la b yb + d 9.
-
Use the framework described in Section 24.3 to construct normalized hybrid bonding orbitals on the central oxygen in O 3 that are derived from 2s and 2p atomic orbitals. The bond angle in ozone is...
-
How can cattle grazing be used to improve elk winter range?
-
A snow-cone machine at an icecream shop costs \(\$ 15,000\). The machine is expected to generate profits of \(\$ 2,500\) each year of its 10-year useful life. At the end of the 10 years, the machine...
-
A small truck is purchased for \($17,000.\) It is expected to be of use to the company for 6 years, after which it will be sold for \($3,500.\) Determine the depreciation deduction and the resulting...
-
1. To develop an understanding of your ethical leadership style 2. To understand how your preferred ethical leadership style relates to other ethical leadership styles Directions 1. Please read the...
-
Consider the following Keynesian closed economy: Consumption \(\quad C=388+0.4(Y-T)-600 r\). Investment \(I=352-400 r\). Government purchases \(G=280\). Taxes \(\quad T=300\). Full-employment output...
-
What is the approximate magnitude of the electric field between the charge center and the ground? A. \(4 \times 10^{4} \mathrm{~V} / \mathrm{m}\) B. \(4 \times 10^{5} \mathrm{~V} / \mathrm{m}\) C....
-
Ducs wallet contains 8 bills of the following denominations: four $5 bills, two $10 bills, one $20 bill, and one $50 bill. If Duc selects two bills at random, determine the probability that he...
-
Element compound homogeneous mixture (heterogeneous mixture) 4) A piece of gold has a mass of 49.75 g. What should the volume be if it is pure gold? Gold has a density of 19.3 g/cm (3 points) D=m/v...
-
Provide a molecular interpretation for each of the following processes: diffusion, thermal conduction, electric conduction, and viscosity.
-
Determine the ratios of (a) The mean speeds, (b) The mean kinetic energies of H 2 molecules and Hg atoms at 20C.
-
The gas-phase association reaction between F 2 and IF 5 is first-order in each of the reactants. The energy of activation for the reaction is 58.6 kJ mol 1 . At 65C the rate constant is 7.84 10 3...
-
The articles of partnership for Pal - Trotter Partnership provide for a salary allowance of $ 5 , 3 0 0 per month for partner Trotter, with the balance of net income to be divided equally. If Trotter...
-
The City of Piglet reported property tax revenues in 2020 in the amount of $10 million. The deferred inflow-property taxes reported in the General Fund's balance sheet was $300,000 on December 31,...
-
Texas Roadhouse is expecting to earn $ 5 million in revenue for the next fiscal year. How much net income would the company need to generate from that revenue to achieve a net profit margin ratio of...

Study smarter with the SolutionInn App


