The following diagram shows the energies of valence molecular orbitals of boron trifluoride. The energies of three
Question:
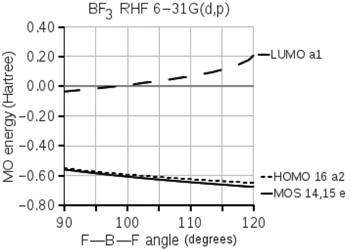
Transcribed Image Text:
BF3 RHF 6-31G(d,p) 0.40 0.20 -LUMO a1 0.00 :-0.20- 5 -0.40 - -0.60 --номо 16 а2 -MOS 14,15 e -0.80 90 100 110 120 F-B-F angle (degrees) MO energy (Hartree)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
BF 3 has 32 electrons which fill up through MOs 14 15 ...View the full answer

Answered By

Muhammad adeel
I am a professional Process/Mechanical engineer having a vast 7 years experience in process industry as well as in academic studies as a instructor. Also equipped with Nebosh IGC and lead auditor (certified).
Having worked at top notch engineering firms, i possess abilities such as designing process equipment, maintaining data sheets, working on projects, technical biddings, designing PFD and PID's etc.
Having worked as an instructor in different engineering institutes and have been involved in different engineering resrearch projects such as refinery equipment designing, thermodynamics, fluid dynamics, chemistry, rotary equipment etc
I can assure a good job within your budget and time deadline
4.90+
52+ Reviews
60+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following diagram shows one-half of a restriction site. (a) Draw the other half. (b) Use heavy arrows () to identify type II cleavage sites that would yield blunt-ended duplex DNA products. (c)...
-
Consider the molecular orbitals of the P2 molecule. Assume that the MOs of di-atomics from the third row of the periodic table are analogous to those from the second row. (a) Which valence atomic...
-
The iodine bromide molecule, IBr, is an inter-halogen compound. Assume that the molecular orbitals of IBr are analogous to the homo nuclear diatomic molecule F2. (a) Which valence atomic orbitals of...
-
Gems Co. uses the indirect method to prepare its statement of cash flows. The following comparative statement of financial position for 2021 and 2022 are presented: At December 31 2022 2021 Property,...
-
Describe the error. cos 60o = opp / hyp = 1 / 2
-
A firm is considering using the Internet to supplement its traditional sales methods. Using data from an industry association, the firm estimates that 1 of every 1,000 Internet hits results in a...
-
The personal training business owned by David Hella made the following cash transactions during the reporting period. Ignore GST. Required (a) Prepare a statement of cash flows using the direct...
-
Frozen North Outfitters Inc. makes thermal clothing for winter sports and outdoor work. It is considering acquiring Downhill Fashions Corp., which manufactures and sells ski clothing. Downhill is...
-
Titleist Inc produces two types of golf balls for the retail consumer. They want to know which golf ball produces the greatest profit and they want to maintain at least $1.50 of profit per golf ball,...
-
Tutors for Rent, Inc., performs adjusting entries every month, but closes its accounts only at year-end. The companys year-end adjusted trial balance dated December 31, 2011, was: a. Prepare an...
-
The energy of the occupied valence MOs of H 2 S is shown as a function of the HSH bond angle. Compared to the analogous diagram, Figure 24.11, for H 2 O, the 2a1 MO energy decreases more as the bond...
-
The density of states (DOS) of pyrite, crystalline FeS 2 (as calculated by Eyert et al, Physical Review B 55 (1998): 6350, is shown next. The highest occupied energy level corresponds to zero energy....
-
The risk-free rate is currently 7%, and the market return is 12%. Assume you are considering the following investments. Investment Beta A ............. 1.5 B ............. 1.0 C ............. 0.75 D...
-
A positively charged solid sphere of radius \(100 \mathrm{~mm}\) has a uniform volume charge density of \(250 \mathrm{nC} / \mathrm{m}^{3}\). Determine the electric field (a) 20 mm, (b) 90 mm, and...
-
An infinitely long positively charged wire with a uniform linear charge density \(+\lambda\) is parallel to the \(y\) axis of a Cartesian coordinate system and passes through the \(x\) axis at...
-
Estimate the mass-transfer coefficient for the dissolution of sodium chloride from a cast sphere, \(1.5 \mathrm{~cm}\) in diameter, if placed in a flowing water stream. The velocity of the \(291...
-
A 1-m square thin plate of solid naphthalene is oriented parallel to a stream of air flowing at \(20 \mathrm{~m} / \mathrm{s}\). The air is at \(330 \mathrm{~K}\) and \(101.3 \mathrm{kPa}\). The...
-
Sketch the graph of each equation in Problems 3-30. \(y=2 x^{2}-3\)
-
Convert each improper fraction to a mixed number. 13/4
-
A regular deposit of $100 is made at the beginning of each year for 20 years. Simple interest is calculated at i% per year for the 20 years. At the end of the 20-year period, the total interest in...
-
The mobilities of H + and Cl at 25C in water are 3.623 10 7 m 2 s 1 V 1 and 7.91 10 8 m 2 s 1 V 1 , respectively. What proportion of the current is carried by the protons in 10 3 m HCl(aq)? What...
-
A space vehicle of internal volume 3.0 m 3 is struck by a meteor and a hole of radius 0.10 mm is formed. If the oxygen pressure within the vehicle is initially 80 kPa and its temperature 298 K, how...
-
A manometer was connected to a bulb containing carbon dioxide under slight pressure. The gas was allowed to escape through a small pinhole, and the time for the manometer reading to drop from 75 cm...
-
The sheet Inventory lists a grocery store's inventory for two months. Calculate the percent change for each item to two decimal places. If the formula gives an error, put "Initial Stock" in the cell....
-
A local manufacturer produces two different products on the same production line. Product A requires .5 hours of labor while product B requires .8 hours of labor. The total labor available in any one...
-
Hampton Company reports the following information for its recent calendar year. Income Statement Data Sales Expenses: Cost of goods sold Salaries expense Depreciation expense Net income Required:...

Study smarter with the SolutionInn App


